regulation
On this blog, I have almost exclusively written about so-called alternative medicine (SCAM), and I intend to continue along these lines. But today, I have to make an exception. The reason is the UK government’s ‘Rwanda Project’. I heard about it this morning while listening to the BBC, and I am too shocked to not write about it.
Already in 2020, it had been reported that the UK home secretary, Priti Patel, had asked officials to look into the possibility of sending all asylum seekers to Ascension, an isolated volcanic British island south of the Equator in the Atlantic Ocean.[1] At the time, the idea was met with much criticism and opposition. Today she is about to announce that a modification of her plan will, in fact, be implemented; our government is about to unveil plans to send ‘illegal migrants’ who reach Britain to Rwanda:
Flying asylum seekers to Rwanda to be processed is “despicable” and “evil”, critics have said as they hit out at government plans expected to be announced on Monday. ITV News has seen a government document that raises issues over the legality of such a policy, as ministers attempt to tackle small boat crossings of the Channel. The government document says the policy would carry the risk of legal challenges but is possible under current legislation should the government wish to push ahead. Boris Johnson is set to argue on Thursday that action is needed to combat the “vile people smugglers” turning the ocean into a “watery graveyard”. Many details of the expected announcement, such as whether it would apply just to those who arrived by what the government calls illegal means, remain unclear. The document seen by ITV News also states that any agreement of this nature would require the government to financially incentivise whichever country it reaches a deal with. Initial estimates had the policy costing in the tens of millions of pounds, but the document says this has been revised to the hundreds of millions of pounds.
The plan sounds utterly cynical, irrational, illegal, and expensive; and again there is an outrage; QC tweeted, for instance, this: “And if an asylum applicant who’s been dumped in Rwanda is then refused, what happens then? The whole thing is the stuff of nightmares. It feels like things are becoming increasingly dystopian as the days pass. Why oh why can’t we have some creative and humane people in charge.”
One might well ask, where does such an inhumane concept come from?
It turns out that it resembles an idea of the Third Reich that was equally baffling and similarly cruel.
Anticipating the German invasion of France in 1940, the German diplomat, Franz Rademacher, developed his Madagascar project, a bizarre plan to rid Europe of all Jews. It envisaged that:
- once defeated, France would agree to make Madagascar available,
- American Jewry would be blackmailed into funding the operation,
- France would be forced to find an alternate home for the 25,000 French inhabitants of Madagascar.
The plan may now sound ridiculous but, in 1940, it was seriously considered by the Nazi leadership. In June of that year, Rademacher, who then was the head of the German Foreign Ministry’s Jewish Department, presented the idea to his boss, Joachim von Ribbentrop, who then explained it to Hitler. Hitler was sufficiently impressed to discuss it with Mussolini. Rademacher wrote that “The approaching victory gives Germany the possibility, and in my view also the duty, of solving the Jewish question in Europe. The desirable solution is: All Jews out of Europe.” [2]
When Reinhard Heydrich, chief of the Reich Main Security Office, learned about the “Madagascar Project”, he highjacked it and swiftly put Adolf Eichmann in charge of it. On August 15, 1940, Eichmann published a pamphlet on the “Madagascar Project.” It envisaged that the deportation of the Jews would be carried out by the British merchant marine, after the anticipated invasion and defeat of the United Kingdom. Two ships per day would then leave Europe for Madagascar, each one carrying 1,500 Jews. In just a few years, Europe would thus become ‘Juden-frei’, free of Jews.
The Madagascar plan envisaged turning Madagascar into a monumental concentration camp under German control. The island is merely 228,900 square miles in size and would not have provided sufficient room for millions. It was therefore understood that many of the prisoners would perish, not least because of the lack of food, housing and healthcare as well as the difficult climate. 
Eventually, the Madagascar plan had to be abandoned. What was decided subsequently with Rademacher’s cooperation about the fate of the Jews, the ‘final solution’, was even more unspeakably outrageously abhorrent.
The main players involved in the Madagascar project did all perish:
- Ribbentrop was sentenced to death at the Nuremberg trials.
- Hitler committed suicide.
- Heydrich was assassinated.
- Eichmann was sentenced to death in Israel.
Only Rademacher survived. After the war, he was imprisoned by British military police who mistook him for ‘small fry’ and promptly released him. In 1952, he was put on trial in West Germany for the murders of Jews he had supervised in Serbia. Yet, aided by a network of Nazi sympathizers, he managed to flee to Syria. The German court then convicted him in his absence and sentenced him to 3 years and 5 months imprisonment. In 1962, an Israeli spy delivered a letter bomb to Rademacher in Syria who, however, remained unharmed. In 1966, Rademacher returned voluntarily to West Germany and was promptly re-arrested. This time, he was sentenced to five and half years, but never actually served a prison sentence. In 1971, the German high court overruled the former judgment and ordered a new trial to take place. However, Rademacher died on 17 March 1973, before it could take place.[3]
I hope that I am wrong and that the UK government’s plan is not similar to the Nazi’s “Madagascar Project”. Maybe it is just a PR stunt of our PM to distract from his very own lawbreaking, and the ‘Rwanda project’ will never be implemented. In any case, we should urgently remind everyone of George Santayana’s wise words: “Those who cannot remember the past are condemned to repeat it.”
[1] https://www.independent.co.uk/news/uk/politics/priti-patel-asylum-ascensi on-island-atlantic-immigration-process-centre-b703625.html
[2] https://www.haaretz.com/jewish/.premium-1940-eichmann-plans-to-deport-jews-to-madagascar-1.5424692
Brite is an herbal energy drink that is currently being marketed aggressively. It is even for sale in one leading UK supermarket. It comes in various flavors the ingredients of which vary slightly.
The pineapple/mango drink, for instance, contains:
- guarana extract,
- green tea extract,
- guayusa extract,
- ashwagandha extract,
- matcha tea,
- ascorbic acid (vitamin C),
- natural caffeine.
The website of the manufacturer tells us that Brite uses ingredients and dosages that are safe and effective, utilising the power of nootropic superfoods organic Matcha, Guarana and Guayusa to provide a long-lasting boost.
Brite is based on peer reviewed, double-blind, placebo-controlled clinical trials and studies that can be found here.
It does not tell us the dosages of the ingredients, and I am puzzled by the claim that the drink is safe. A quick search seems to cast considerable doubt on it.
_____________________________
Guarana (Paullinia cupana) is a plant from the Amazon region with a high content of bioactive compounds. It is by no means free of adverse effects. It is known to interact with:
- armodafinil
- caffeine
- dexmethylphenidate
- dextroamphetamine
- green tea
- lisdexamfetamine
- methamphetamine
- methylenedioxymethamphetamine
- methylphenidate
- modafinil
- phentermine
- yohimbine
And it can cause the following adverse effects:
- Abdominal spasms (from overdose)
- Agitation
- Anxiety
- Convulsions
- Delirium
- Dependence
- Diarrhea
- Dizziness
- Fast heart rate
- Gastrointestinal (GI) upset
- Headache
- High blood pressure (hypertension)
- High blood sugar (hyperglycemia)
- Increased respiration
- Increased urination
- Insomnia
- Irregular heartbeats (arrhythmias)
- Irritability
- Muscle spasms
- Nausea/vomiting
- Nervousness
- Painful urination (from overdose)
- Rapid breathing
- Restlessness
- Ringing in the ears (tinnitus)
- Stomach cramps or irritation
- Tremors
- Withdrawal symptoms
Green tea is made from the leaves of the Camellia sinensis plant. It can cause the following adverse effects:
- headache,
- nervousness,
- sleep problems,
- vomiting,
- diarrhea,
- irritability,
- irregular heartbeat,
- tremor,
- heartburn,
- dizziness,
- ringing in the ears,
- convulsions,
- confusion.
Guayusa is a plant native to the Amazon rainforest that contains plenty of caffeine. Its adverse effects include:
- High Blood Pressure
- Rapid Heartbeat
- Anxiety
- Jitters
- Energy Crashes
- Insomnia
- Headaches
- Upset Stomach
Ashwagandha is a plant from India; the root and berry are used in Ayurvedic medicine. Its adverse effects include:
- stomach upset,
- diarrhea,
- vomiting.
Matcha tea also contains a high amount of caffeine. It is associated with the following adverse effects:
- nervousness,
- irritability,
- dizziness,
- anxiety,
- digestive disorders such as irritable bowel syndrome, or diarrhea,
- sleeping disorders,
- cardiac arrhythmia.
Caffeine is a chemical found in coffee, tea, cola, guarana, mate, and other products. Adverse effects include:
- insomnia,
- nervousness,
- restlessness,
- stomach irritation,
- nausea and vomiting,
- increased heart rate and respiration,
- headache,
- anxiety,
- agitation,
- chest pain,
- ringing in the ears.
A case report documented a case of myocardial infarction in a 25-year-old man who presented to the emergency department with chest pain. The patient had been consuming massive quantities of caffeinated energy drinks daily for the past week. This case report and previously documented studies support a possible connection between caffeinated energy drinks and myocardial infarction.
________________________
Yes, the adverse effects are predominantly (but not exclusively) caused by high doses. Yet, the claim that Brite is safe should nevertheless be taken with a very large pinch of salt. If I like the taste of the drink and thus consume a few bottles per day, the dosages of the ingredients would surely be high!
And what about the claim that it is effective? Here the pinch of salt must be even larger, I am afraid. I could not find a single trial that confirmed the notion. For backing up their claims, the manufacturers offer a few references, but if you look them up, you will find that they were not done with the mixture of ingredients contained in Brite.
So, what is the conclusion?
Based on the evidence that I have seen, the herbal drink ‘Brite’ has not been shown to be an effective nootropic. In addition, there are legitimate concerns about the safety of the product. I for one will therefore not purchase the (rather expensive) drink.
The new issue of the BMJ carries an article on acupuncture that cries out for a response. Here, I show you the original article followed by my short comments. For clarity, I have omitted the references from the article and added references that refer to my comments.
_________________________________________
Conventional allopathic medicine [1]—medications and surgery [2] used in conventional systems of medicine to treat or prevent disease [3]—is often expensive, can cause side effects and harm, and is not always the optimal treatment for long term conditions such as chronic pain [4]. Where conventional treatments have not been successful, acupuncture and other traditional and complementary medicines have potential to play a role in optimal patient care [5].
According to the World Health Organisation (WHO) 2019 global report, acupuncture is widely used across the world. [6] In some countries acupuncture is covered by health insurance and established regulations. [7] In the US, practitioners administer over 10 million acupuncture treatments annually. [6] In the UK, clinicians administer over 4 million acupuncture treatments annually, and it is provided on the NHS. [6]
Given the widespread use of acupuncture as a complementary therapy alongside conventional medicine, there has been an increase in global research interest and funding support over recent decades. In 2009, the European Commission launched a Good Practice in Traditional Chinese Medicine Research (GP-TCM) funding initiative in 19 countries. [7] The GP-TCM grant aimed to investigate the safety and efficacy of acupuncture as well as other traditional Chinese medicine interventions.
In China, acupuncture is an important focus of the national research agenda and receives substantial research funding. [8] In 2016, the state council published a national strategy supporting universal access to acupuncture by 2020. China has established more than 79 evidence-based traditional Chinese medicine or integrative medicine research centers. [9]
Given the broad clinical application and rapid increase in funding support for acupuncture research, researchers now have additional opportunities to produce high-quality studies. However, for this to be successful, acupuncture research must address both methodological limitations and unique research challenges.
This new collection of articles, published in The BMJ, analyses the progress of developing high quality research studies on acupuncture, summarises the current status, and provides critical methodological guidance regarding the production of clinical evidence on randomised controlled trials, clinical practice guidelines and health economic evidence. It also assesses the number and quality of systematic reviews of acupuncture. [10] We hope that the collection will help inform the development of clinical practice guidelines, health policy, and reimbursement decisions. [11]
The articles document the progress of acupuncture research. In our view, the emerging evidence base on the use of acupuncture warrants further integration and application of acupuncture into conventional medicine. [12] National, regional, and international organisations and health systems should facilitate this process and support further rigorous acupuncture research.
Footnotes
This article is part of a collection funded by the special purpose funds for the belt and road, China Academy of Chinese Medical Sciences, National Natural Science Foundation of China, the National Center for Complementary and Integrative Health, the Innovation Team and Talents Cultivation Program of the National Administration of Traditional Chinese Medicine, the Special Project of “Lingnan Modernization of Traditional Chinese Medicine” of the 2019 Guangdong Key Research and Development Program, and the Project of First Class Universities and High-level Dual Discipline for Guangzhou University of Chinese Medicine. The BMJ commissioned, peer reviewed, edited, and made the decision to publish. Kamran Abbasi was the lead editor for The BMJ. Yu-Qing Zhang advised on commissioning for the collection, designed the topic of the series, and coordinated the author teams. Gordon Guyatt provided valuable advice and guidance. [13]
1. Allopathic medicine is the term Samuel Hahnemann coined for defaming conventional medicine. Using it in the first sentence of the article sets the scene very well.
2. Medicine is much more than ‘medications and surgery’. To imply otherwise is a strawman fallacy.
3. What about rehabilitation medicine?
4. ‘Conventional medicine is not always the optimal treatment’? This statement is very confusing and wrong. It is true that conventional medicine is not always effective. However, it is by definition the best we currently have and therefore it IS optimal.
5. Another fallacy: non sequitur
6. Another fallacy: appeal to popularity.
7. Yet another fallacy: appeal to authority.
8. TCM is heavily promoted by China not least because it is a most lucrative source of income.
9. Several research groups have shown that 100% of acupuncture research coming out of China report positive results. This casts serious doubt on the reliability of these studies (see, for instance, here, here, and here).
10. It has been noted that more than 80 percent of clinical data from China is fabricated.
11. Based on the points raised above, it seems to me that the collection’s aim is not to provide objective information but uncritical promotion.
12. I find it telling that the authors do not even consider the possibility that rigorous research might demonstrate that acupuncture cannot generate more good than harm.
13. This statement essentially admits that the series of articles constitutes paid advertising for TCM. The BMJ’s peer-review process must have been less than rigorous in this case.
Conversion therapy has been banned last week in Canada. These therapies – also known as sexual orientation change effort (SOCE), reparative therapy, reintegrative therapy, reorientation therapy, ex-gay therapy, and gay cure – rely on the assumption that sexual orientation can be changed, an idea long discredited by major medical associations in the US, the UK, France, and elsewhere. The new law makes “providing, promoting, or advertising conversion therapy” a criminal offense. It will also be an offense to profit from the provision of conversion therapy. In addition, the bill states a person cannot remove a “child from Canada with the intention that the child undergo conversion therapy outside Canada.” Prime Minister Justin Trudeau hailed the law’s Royal Assent: “It’s official: Our government’s legislation banning the despicable and degrading practice of conversion therapy has received Royal Assent — meaning it is now law.”
Conversion therapy is the attempt to change an individual’s sexual or gender identity by psychological, medical, or surgical interventions. Often, informed consent is insufficient or lacking. In conventional medicine, numerous treatments have been tried for this purpose, some of them dangerous and all of them ineffective. In alternative medicine, approaches that have been advocated include:
- Homeopathy (see below),
- Hypnotherapy,
- Spiritual healing,
- Prayer,
- Eye Movement Desensitization,
- Rebirthing,
- and others.
- Faith-based organizations or leaders
- Licensed healthcare professionals
- Unlicensed healthcare professionals
As previously reported, the German ‘Association of Catholic Doctors’ claimed that homeopathic remedies can cure homosexuality. Specifically, they advised that ‘…the working group ‘HOMEOPATHY’ of the Association notes homeopathic therapy options for homosexual tendencies…repertories contain special rubrics pointing to characteristic signs of homosexual behavior, including sexual peculiarities such as anal intercourse. And a homeopathic remedy called ‘Dr. Reckeweg R20 Glandular Drops for Women’ was claimed to treat “lesbian tendencies.” The product is “derived and potentised from fetal tissues.”
Several countries are now in the process of banning conversion therapy. France has already banned it and so has Germany. The UK government intends to introduce a legislative ban on the practice of conversion therapy. The consultation on how to best do this is open until 4 February 2022.
The complex links between so-called alternative medicine (SCAM) and the pandemic have been a regular subject on this blog. Here is more:
This study investigated if people’s response to the official recommendations during the COVID-19 pandemic is associated with conspiracy beliefs related to COVID-19, a distrust in the sources providing information on COVID-19, and an endorsement of SCAM.
The sample consisted of 1325 Finnish adults who filled out an online survey advertised on Facebook. Structural regression analysis was used to investigate whether:
1) conspiracy beliefs, a distrust in information sources, and endorsement of SCAM predict people’s response to the non-pharmaceutical interventions (NPIs) implemented by the government during the COVID-19 pandemic,
2) conspiracy beliefs, a distrust in information sources, and endorsement of CAM are related to people’s willingness to take a COVID-19 vaccine.
The results indicate that individuals with more conspiracy beliefs and lower trust in information sources were less likely to have a positive response to the NPIs. Individuals with less trust in information sources and more endorsement of SCAM were more unwilling to take a COVID-19 vaccine. Distrust in information sources was the strongest and most consistent predictor in all models. In addition, the analyses revealed that some of the people who respond negatively to the NPIs also have a lower likelihood to take the vaccine. This association was partly related to lower trust in information sources.
The authors concluded that distrusting the establishment to provide accurate information, believing in conspiracy theories, and endorsing treatments and substances that are not part of conventional medicine, are all associated with a more negative response to the official guidelines during COVID-19. How people respond to the guidelines, however, is more strongly and consistently related to the degree of trust they feel in the information sources than to their tendency to hold conspiracy beliefs or endorse CAM. These findings highlight the need for governments and health authorities to create communication strategies that build public trust.
I also believe that these findings highlight the urgent need for improvements in education. In my view, it should start at school and continue into adult life. It should focus on a better understanding of science and – crucially – on the ability to differentiate facts from fiction and conspiracies.
The bad news for German homeopathy just keeps on coming. As I reported, recent events must be depressing for homeopaths, e.g.:
- A recent investigation disclosed that the German homeopathy cult is at the heart of Germany’s anti-vax movement.
- In July 2021, a German court ruled against a homeopathic HCG remedy.
- In 2020, a German study failed to show that homeopathy is cost-effective.
- In 2019, the attempt by a German homeopathy manufacturer to silence critics backfired spectacularly.
- About 10 years ago, 5 German firms clubbed together supporting a journalist at the tune of Euro 30 000/year to defame me (at the time I was one of the few who regularly informed the public about the evidence on homeopathy). The plot was discovered and published whereupon the journalist committed suicide.
After heated debates in the run-up, the Bavarian Medical Association decided yesterday to ditch the postgraduate education program in homeopathy for its doctors. This means that, of the 17 regional medical associations in Germany, 12 have now discontinued their further education efforts in homeopathy. The ones that have not yet done so are:
- Baden-Württemberg,
- Rhineland-Palatinate,
- Saxony,
- Thuringia,
- Westphalia-Lippe.
In the past months, homeopaths had collected 11,597 signatures in favor of maintaining the additional qualification of homeopathy. The ~ 400 doctors in Bavaria, who have acquired ‘homeopathy’ as an additional title, will be permitted to continue to use it.
The spokesperson of the Information Network Homeopathy, Dr. Christian Lübbers, welcomed the decision of the Medical Association. It was a “landslide victory for patient safety”, he said. The Bavarian regional chairman of the German Central Association of Homeopathic Doctors, Dr. Ulf Riker, regretted the outcome of the vote and added: “We will consider legal steps very seriously.” I would advise against such a step which would only render homeopaths more ridiculous than they already are.
Yes, it’s bad news for German homeopaths – very bad news indeed. Of course, homeopathy fans will claim that it is all a sinister conspiracy against them. Sadly, they are unable to realize that the only driving force behind the long-overdue decline of German homeopathy is the evidence: HOMEOPATHY DOES NOT WORK BEYOND PLACEBO and therefore it has no place in the evidence-based medicine of the 21st century.
This article from AP News caught my attention. Here it is (I haven’t changed a word):
The flashy postcard, covered with images of syringes, beckoned people to attend Vax-Con ’21 to learn “the uncensored truth” about COVID-19 vaccines.
Participants traveled from around the country to a Wisconsin Dells resort for a sold-out convention that was, in fact, a sea of misinformation and conspiracy theories about vaccines and the pandemic. The featured speaker was the anti-vaccine activist who appeared in the 2020 movie “Plandemic,” which pushed false COVID-19 stories into the mainstream. One session after another discussed bogus claims about the health dangers of mask wearing and vaccines.
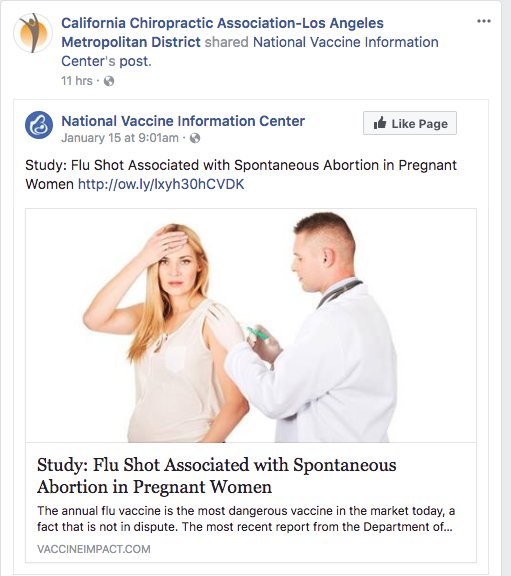
The convention was organized by members of a profession that has become a major purveyor of vaccine misinformation during the pandemic: chiropractors.
At a time when the surgeon general says misinformation has become an urgent threat to public health, an investigation by The Associated Press found a vocal and influential group of chiropractors has been capitalizing on the pandemic by sowing fear and mistrust of vaccines.
They have touted their supplements as alternatives to vaccines, written doctor’s notes to allow patients to get out of mask and immunization mandates, donated large sums of money to anti-vaccine organizations and sold anti-vaccine ads on Facebook and Instagram, the AP discovered. One chiropractor gave thousands of dollars to a Super PAC that hosted an anti-vaccine, pro-Donald Trump rally near the U.S. Capitol on Jan. 6.
They have also been the leading force behind anti-vaccine events like the one in Wisconsin, where hundreds of chiropractors from across the U.S. shelled out $299 or more to attend. The AP found chiropractors were allowed to earn continuing education credits to maintain their licenses in at least 10 states.
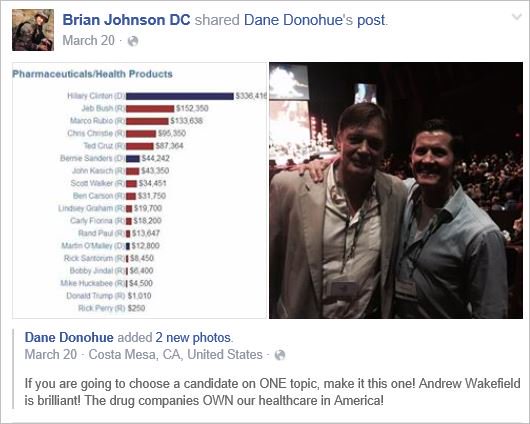
On this blog, I have often discussed that chiropractors tend to be anti-vax. It all goes back to their founding father, DD Palmer, who famously wrote:
- Vaccination and inoculation are pathological; chiropractic is physiological,
- and who in 1894, published his views on smallpox vaccination: ‘…the monstrous delusion … fastened on us by the medical profession, enforced by the state boards, and supported by the mass of unthinking people …’
- and who stated in 1896 that keeping tissue healthy is therefore the best prevention against infections; and this is best achieved by magnetic healing.
But that’s long ago! We are not like that anymore! … say the chiros of today.
Do you believe them?
If so, you might want to read this article by Jann Bellamy. Or alternatively, just look at some of my finds from the Internet:
Cannabis seems often to be an emotional subject where more heat than light is generated. Does it work for chronic pain? This cannot be such a difficult question to answer definitively. Yet, systematic reviews have provided conflicting results due, in part, to limitations of analytical approaches and interpretation of findings.
A new systematic review is therefore both necessary and welcome. It aimed at determining the benefits and harms of medical cannabis and cannabinoids for chronic pain. Included were all randomised clinical trials of medical cannabis or cannabinoids versus any non-cannabis control for chronic pain at ≥1-month follow-up.
A total of 32 trials with 5174 adult patients were included, 29 of which compared medical cannabis or cannabinoids with placebo. Medical cannabis was administered orally (n=30) or topically (n=2). Clinical populations included chronic non-cancer pain (n=28) and cancer-related pain (n=4). Length of follow-up ranged from 1 to 5.5 months.
Compared with placebo, non-inhaled medical cannabis probably results in a small increase in the proportion of patients experiencing at least the minimally important difference (MID) of 1 cm (on a 10 cm visual analogue scale (VAS)) in pain relief (modelled risk difference (RD) of 10% (95% confidence interval 5% to 15%), based on a weighted mean difference (WMD) of −0.50 cm (95% CI −0.75 to −0.25 cm, moderate certainty)). Medical cannabis taken orally results in a very small improvement in physical functioning (4% modelled RD (0.1% to 8%) for achieving at least the MID of 10 points on the 100-point SF-36 physical functioning scale, WMD of 1.67 points (0.03 to 3.31, high certainty)), and a small improvement in sleep quality (6% modelled RD (2% to 9%) for achieving at least the MID of 1 cm on a 10 cm VAS, WMD of −0.35 cm (−0.55 to −0.14 cm, high certainty)). Medical cannabis taken orally does not improve emotional, role, or social functioning (high certainty). Moderate certainty evidence shows that medical cannabis taken orally probably results in a small increased risk of transient cognitive impairment (RD 2% (0.1% to 6%)), vomiting (RD 3% (0.4% to 6%)), drowsiness (RD 5% (2% to 8%)), impaired attention (RD 3% (1% to 8%)), and nausea (RD 5% (2% to 8%)), but not diarrhoea; while high certainty evidence shows greater increased risk of dizziness (RD 9% (5% to 14%)) for trials with <3 months follow-up versus RD 28% (18% to 43%) for trials with ≥3 months follow-up; interaction test P=0.003; moderate credibility of subgroup effect).
The authors concluded that moderate to high certainty evidence shows that non-inhaled medical cannabis or cannabinoids results in a small to very small improvement in pain relief, physical functioning, and sleep quality among patients with chronic pain, along with several transient adverse side effects, compared with placebo.
This is a high-quality review. Its findings will disappoint the many advocates of cannabis as a therapy for chronic pain management. The bottom line, I think, seems to be that cannabis works but the effect is not very powerful, while we have treatments for managing chronic pain that are both more effective and arguably less risky. So, its place in clinical routine is debatable.
PS
Cannabis is, of course, a herbal remedy and therefore belongs to so-called alternative medicine (SCAM). Yet, I am aware that the medical cannabis preparations used in most studies are based on single cannabinoids which makes them conventional medicines.
The General Chiropractic Council (GCC) regulates chiropractors in the UK, Isle of Man, and Gibraltar to ensure the safety of patients undergoing chiropractic treatment. The GCC sets the standards of chiropractic practice and professional conduct that all chiropractors must meet.

By providing a lengthy ruling in the case of the late John Lawler and his chiropractor, Arlene Scholten, the GCC has recently established new standards for chiropractors working in the UK, Isle of Man, and Gibraltar (see also today’s article in The Daily Mail). If I interpret the GCC’s ruling correctly, a UK chiropractor is henceforth allowed to do all of the following things without fearing to get reprimanded, as long as he or she produces evidence that the deeds were done not with malicious intentions but in a state of confusion and panic:
- Treat a patient with treatments that are contraindicated.
- Fail to obtain informed consent.
- Pose as a real doctor without informing the patient that the practitioner is just a chiropractor who has never been near a medical school.
- Cause the death of a patient by treatment to the neck.
- Administer first aid in a way that makes matters worse.
- Tell lies to the ambulance men who consequently failed to employ a method of transport that would save the patient’s life.
- Keep inaccurate patient records that conceal what treatments were administered.
In previous years, the job of a chiropractor had turned out to be demanding, difficult, and stressful. This was due not least to the GCC’s professional standards which UK chiropractors were obliged to observe. The code of the GCC stated prominently that “our overall purpose is to protect the public.”
All this is now a thing of the past.
The new ruling changed everything. Now, UK chiropractors can relax and can happily pursue their true devotion, namely to keep their bank manager happy, while not worrying too much about the welfare and health of their patients.
In the name of all UK chiropractors, I herewith express my thanks to the GCC for unashamedly protecting first and foremost the interests of their members, while tacitly discarding medical ethics and evidently not protecting the public.
MAKE CHIROPRACTIC GREAT AGAIN!
This retrospective electronic medical record data analysis compared the characteristics and outcomes of drug-induced liver injury (DILI) caused by paracetamol and non-paracetamol medications, particularly herbal and dietary supplements. Adults admitted with DILI to the Gastroenterology and Liver Centre at the Royal Prince Alfred Hospital, Sydney (a quaternary referral liver transplantation centre), 2009-2020 were included. The 90-day transplant-free survival and the drugs implicated as causal agents in DILI were extracted from the records.
A total of 115 patients with paracetamol-related DILI and 69 with non-paracetamol DILI were admitted to our centre. The most frequently implicated non-paracetamol medications were:
- antibiotics (19, 28%),
- herbal and dietary supplements (15, 22%),
- anti-tuberculosis medications (6, 9%),
- anti-cancer medications (5, 7%).
The number of non-paracetamol DILI admissions was similar across the study period, but the proportion linked with herbal and dietary supplements increased from 2 of 11 (15%) during 2009-11 to 10 of 19 (47%) during 2018-20 (linear trend: P = 0.011). Despite higher median baseline model for end-stage liver disease (MELD) scores, 90-day transplant-free survival for patients with paracetamol-related DILI was higher than for patients with non-paracetamol DILI (86%; 95% CI, 79-93% v 71%; 95% CI, 60-82%) and herbal and dietary supplement-related cases (59%; 95% CI, 34-85%). MELD score was an independent predictor of poorer 90-day transplant-free survival in both paracetamol-related (per point increase: adjusted hazard ratio [aHR], 1.19; 95% CI, 1.09-3.74) and non-paracetamol DILI (aHR, 1.24; 95% CI, 1.14-1.36).
The authors concluded that, in our single centre study, the proportion of cases of people hospitalised with DILI linked with herbal and dietary supplements has increased since 2009. Ninety-day transplant-free survival for patients with non-paracetamol DILI, especially those with supplement-related DILI, is poorer than for those with paracetamol-related DILI.
A co-author of the paper, specialist transplant hepatologist Dr Ken Liu, was quoted in the Guardian saying he felt compelled to conduct the study because he was noticing more patients with liver injuries from drugs not typically associated with liver harm. “I was starting to see injury in patients admitted with liver injury after using bodybuilding supplements for males or weight loss supplements in females,” he said. “I just decided I better do a study on it to see if my hunch that more of these substances were causing these injuries was true.”
Liu and his colleagues said there needed to be more rigorous regulatory oversight for supplements and other alternative and natural therapies. They also noticed almost half the patients with supplement-induced severe liver injury had non-European ethnic backgrounds. Liu said more culturally appropriate community education about the risks of supplements was needed.
Dr Ken Harvey, public health physician and president of Friends of Science in Medicine, said it was important to note that Liu’s study only examined the most severe cases of supplement-induced liver harm and that the actual rate of harm was likely much higher. “The study only examines severe cases admitted to a specialised liver unit; they cannot be extrapolated to the overall incidence of complementary medicine associated liver injury in Australia,” Harvey said.
The Royal Australian College of General Practitioners, Choice, Friends of Science in Medicine and others have called for an educational statement on the pack and promotional material of medicines making traditional claims, for example saying “This product is based on traditional beliefs and not modern scientific evidence”.
“This was opposed by industry and the TGA,” Harvey said. “But is still needed.”