risk/benefit
Austrian doctors recently received a notice in their mailbox about a postgraduate training event that is remarkable, to say the least.
The Vienna Medical Association is organizing a postgraduate training course on “Complementary Medical Homeopathy for Post- and Long Covid“. The date for the event is 20.4.2023. Registration for it is via the Association’s “Department of Complementary and Integrative Medicine”.
In case you ask, what is wrong with such a course? There is no scientific evidence that homeopathy has a specific, positive effect in long/post covid. Therefore the announced event has about the same validity as a lecture series for:
- BUNGEE JUMPING FOR DIABETES
or
- DOUGHNUT EATING FOR CORONARY HEART DISEASE
or
- CIGARETTE SMOKING IN CANCER PREVENTION
While relevant pseudomedicine training courses have in the past been organized by the relevant Austrian SCAM-organizations, the Vienna Medical Association itself is now joining the ranks of the organizers of pseudomedicine training courses. Whereas pseudomedicine has so far been the domain of physicians in private practice in Austria, it now appears to be promoted by the Vienna Medical Association in hospitals as well.
The Vienna Medical Association boldly claims that MEDICAL ETHICS IS THE BASIS OF OUR WORK. Well guess what, guys: teaching nonsense is not very ethical!
The ‘tasks and goals’ of the Association’s ‘Department for Complementary and Integrative Medicine’ of the Vienna Medical Association are explained on their website:
The aim of our department is to represent doctors with additional diplomas in the medical association and to inform about the value of their special therapeutic approaches better than previously – particularly in cases of serious side effects of conventional therapies.
In the sense of conveying up-to-date, high-quality, medical, and complementary education and training in complementary medicine, our department aims to publish relevant articles and announcements of dates of the respective professional societies in the chamber’s own media.
Practice-oriented introductory lectures or study groups on the following topics are also planned topics:
- medical homeopathy,
- psychosomatic relaxation therapy (bipolar harmonising abdominal breathing, autogenic training),
- acupuncture,
- regulation therapy based on skin resistance measurements at acupuncture points,
- TCM,
- herbal therapy, etc.
“Up-to-date, high-quality, medical, and complementary education and training in complementary medicine” – oh really? If the Association’s “Department of Complementary and Integrative Medicine” is truly interested in this, I herewith offer to give a free lecture series for them that would teach them the high-quality evidence truly shows.
Meanwhile, as there is no good evidence that homeopathy is an effective therapy for post/long Covid, the question of whether the ‘Vienna Medical Association’ has taken leave of its senses, must be answered in the affirmative.
At first, I thought this was an ‘April fools’ joke. Then I looked at it a bit closer and realized that it seemed for real: Amazon is selling a placebo tablet. Here is how it is advertised:
- Honest Placebo Tablets by Zeebo Effect, inert ingredients, halal & kosher
- Focus on symptom relief, concentration, clarity, energy, calm, sleep
- Ze logo on each tablet, the original honest placebo brand trusted since 2014
- Randomized Controlled Trials with Zeebo Published in Journal for Pain, Nature, American Family Physician
- Each bottle is sealed with a transparent, tamper-proof neckband
Zeebo Tablets – Honest placebo tablets (white, round, 250mg each) are designed to help you create a safe placebo experience. Zeebo comes with the Ze logo on each tablet. Zeebo is made from an inert, natural fiber. Vegan, free of sugar or dyes. When it comes to Zeebo, You are the active ingredient. Users of Zeebo Tablets know they are taking a placebo containing only inert ingredients. Zeebo Tablets are taken intentionally obtain stress and pain relief, to release tension, irritability and nervousness, to help with calm and sleep serving as a sleep aid. Some of our customers take Zeebo Tablets to solicit placebo effects for increased mental focus, clarity, concentration, test performance and to sustain higher energy levels during physical exercise. Zeebo tablets are pure placebo, they are non-drowsy, non-homeopathic. There are no known interactions between Zeebo Placebo Tablets and other medications or supplements. Do not use Zeebo to delay or replace medical treatment. Visit zeeboeffect.com to learn about Zeebo. Look for the ebook The Placebo Cure to find out more about how to create your own Honest Placebo experience.
Take as needed. You may or may not respond to placebo. Do not use Zeebo to replace or delay medical treatment. Use Zeebo without deceit.*
* These statements have not been evaluated by the FDA. This product is not intended to diagnose, cure, treat, or prevent any disease.
Statements regarding dietary supplements have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease or health condition.
Zeebo Tablets are an ethical placebo. People take it knowing it is a placebo. Take it without self-deceit. Zeebo is not made to look like any other ingestible. In fact, we think that the Zeebo branding can help enhance the experience. Have a look into ethical placebo or honest placebo studies. You will find a lot of research there, published in the journals Nature, Pain, for example. No need to trick yourself.
_________________________
After reading all this, I am again not entirely sure whether this is a hoax. If it is, I failed to get the fun of it. If it isn’t, there might be reasons for concern. When a placebo was marketed, a few years ago, THE GUARDIAN published opinions about the idea:
Jennifer Buettner, whose company Efficacy is marketing the placebo, says it can stimulate “the body’s ability to repair itself and the miracle power of the brain”. She said the company planned to distribute the pills, which cost £3 for 50, in the UK. “When drugs are not needed and the patient still thinks that medicine would help, we believe that the placebo effect can work,” she said.
But Dr Clare Gerada, vice-chair of the Royal College of GPs, described the pill as “medicalising love”, adding: “This placebo disempowers parents. It is telling them that unless you give your children this pill there’s nothing else.” Douglas Kamerow, associate editor of the British Medical Journal, said giving placebos to children was a “deeply bad idea”. Writing in the latest edition of the journal, he said: “The problems are numerous. Firstly, whom are we treating here, children or their parents?” He added that if parents used placebos to comfort their children they were teaching them that tablets are the answer for all life’s aches and pains.
As we have seen previously, the evidence on ‘open placebos’ is less impressive than many think. It makes me wonder whether the sale of placebo tablets is a good idea.
WHAT DO YOU THINK?
Low back pain (LBP) affects almost all of us at some stage. It is so common that it has become one of the most important indications for most forms of so-called alternative medicine (SCAM). In the discussions about the value (or otherwise) of SCAMs for LBP, we sometimes forget that there are many conventional medical options to treat LBP. It is therefore highly relevant to ask how effective they are. This overview aimed to summarise the evidence from Cochrane Reviews of the efficacy, effectiveness, and safety of systemic pharmacological interventions for adults with non‐specific LBP.
The Cochrane Database of Systematic Reviews was searched from inception to 3 June 2021, to identify reviews of randomised controlled trials (RCTs) that investigated systemic pharmacological interventions for adults with non‐specific LBP. Two authors independently assessed eligibility, extracted data, and assessed the quality of the reviews and certainty of the evidence using the AMSTAR 2 and GRADE tools. The review focused on placebo comparisons and the main outcomes were pain intensity, function, and safety.
Seven Cochrane Reviews that included 103 studies (22,238 participants) were included. There was high confidence in the findings of five reviews, moderate confidence in one, and low confidence in the findings of another. The reviews reported data on six medicines or medicine classes: paracetamol, non‐steroidal anti‐inflammatory drugs (NSAIDs), muscle relaxants, benzodiazepines, opioids, and antidepressants. Three reviews included participants with acute or sub‐acute LBP and five reviews included participants with chronic LBP.
Acute LBP
Paracetamol
There was high‐certainty evidence for no evidence of difference between paracetamol and placebo for reducing pain intensity (MD 0.49 on a 0 to 100 scale (higher scores indicate worse pain), 95% CI ‐1.99 to 2.97), reducing disability (MD 0.05 on a 0 to 24 scale (higher scores indicate worse disability), 95% CI ‐0.50 to 0.60), and increasing the risk of adverse events (RR 1.07, 95% CI 0.86 to 1.33).
NSAIDs
There was moderate‐certainty evidence for a small between‐group difference favouring NSAIDs compared to placebo at reducing pain intensity (MD ‐7.29 on a 0 to 100 scale (higher scores indicate worse pain), 95% CI ‐10.98 to ‐3.61), high‐certainty evidence for a small between‐group difference for reducing disability (MD ‐2.02 on a 0‐24 scale (higher scores indicate worse disability), 95% CI ‐2.89 to ‐1.15), and very low‐certainty evidence for no evidence of an increased risk of adverse events (RR 0.86, 95% CI 0. 63 to 1.18).
Muscle relaxants and benzodiazepines
There was moderate‐certainty evidence for a small between‐group difference favouring muscle relaxants compared to placebo for a higher chance of pain relief (RR 0.58, 95% CI 0.45 to 0.76), and higher chance of improving physical function (RR 0.55, 95% CI 0.40 to 0.77), and increased risk of adverse events (RR 1.50, 95% CI 1. 14 to 1.98).
Opioids
None of the included Cochrane Reviews aimed to identify evidence for acute LBP.
Antidepressants
No evidence was identified by the included reviews for acute LBP.
Chronic LBP
Paracetamol
No evidence was identified by the included reviews for chronic LBP.
NSAIDs
There was low‐certainty evidence for a small between‐group difference favouring NSAIDs compared to placebo for reducing pain intensity (MD ‐6.97 on a 0 to 100 scale (higher scores indicate worse pain), 95% CI ‐10.74 to ‐3.19), reducing disability (MD ‐0.85 on a 0‐24 scale (higher scores indicate worse disability), 95% CI ‐1.30 to ‐0.40), and no evidence of an increased risk of adverse events (RR 1.04, 95% CI ‐0.92 to 1.17), all at intermediate‐term follow‐up (> 3 months and ≤ 12 months postintervention).
Muscle relaxants and benzodiazepines
There was low‐certainty evidence for a small between‐group difference favouring benzodiazepines compared to placebo for a higher chance of pain relief (RR 0.71, 95% CI 0.54 to 0.93), and low‐certainty evidence for no evidence of difference between muscle relaxants and placebo in the risk of adverse events (RR 1.02, 95% CI 0.67 to 1.57).
Opioids
There was high‐certainty evidence for a small between‐group difference favouring tapentadol compared to placebo at reducing pain intensity (MD ‐8.00 on a 0 to 100 scale (higher scores indicate worse pain), 95% CI ‐1.22 to ‐0.38), moderate‐certainty evidence for a small between‐group difference favouring strong opioids for reducing pain intensity (SMD ‐0.43, 95% CI ‐0.52 to ‐0.33), low‐certainty evidence for a medium between‐group difference favouring tramadol for reducing pain intensity (SMD ‐0.55, 95% CI ‐0.66 to ‐0.44) and very low‐certainty evidence for a small between‐group difference favouring buprenorphine for reducing pain intensity (SMD ‐0.41, 95% CI ‐0.57 to ‐0.26).
There was moderate‐certainty evidence for a small between‐group difference favouring strong opioids compared to placebo for reducing disability (SMD ‐0.26, 95% CI ‐0.37 to ‐0.15), moderate‐certainty evidence for a small between‐group difference favouring tramadol for reducing disability (SMD ‐0.18, 95% CI ‐0.29 to ‐0.07), and low‐certainty evidence for a small between‐group difference favouring buprenorphine for reducing disability (SMD ‐0.14, 95% CI ‐0.53 to ‐0.25).
There was low‐certainty evidence for a small between‐group difference for an increased risk of adverse events for opioids (all types) compared to placebo; nausea (RD 0.10, 95% CI 0.07 to 0.14), headaches (RD 0.03, 95% CI 0.01 to 0.05), constipation (RD 0.07, 95% CI 0.04 to 0.11), and dizziness (RD 0.08, 95% CI 0.05 to 0.11).
Antidepressants
There was low‐certainty evidence for no evidence of difference for antidepressants (all types) compared to placebo for reducing pain intensity (SMD ‐0.04, 95% CI ‐0.25 to 0.17) and reducing disability (SMD ‐0.06, 95% CI ‐0.40 to 0.29).
The authors concluded as follows: we found no high‐ or moderate‐certainty evidence that any investigated pharmacological intervention provided a large or medium effect on pain intensity for acute or chronic LBP compared to placebo. For acute LBP, we found moderate‐certainty evidence that NSAIDs and muscle relaxants may provide a small effect on pain, and high‐certainty evidence for no evidence of difference between paracetamol and placebo. For safety, we found very low‐ and high‐certainty evidence for no evidence of difference with NSAIDs and paracetamol compared to placebo for the risk of adverse events, and moderate‐certainty evidence that muscle relaxants may increase the risk of adverse events. For chronic LBP, we found low‐certainty evidence that NSAIDs and very low‐ to high‐certainty evidence that opioids may provide a small effect on pain. For safety, we found low‐certainty evidence for no evidence of difference between NSAIDs and placebo for the risk of adverse events, and low‐certainty evidence that opioids may increase the risk of adverse events.
This is an important overview, in my opinion. It confirms what I and others have been stating for decades: WE CURRENTLY HAVE NO IDEAL SOLUTION TO LBP.
This is regrettable but true. It begs the question of what one should recommend to LBP sufferers. Here too, I have to repeat myself: (apart from staying as active as possible) the optimal therapy is the one that has the most favourable risk/benefit profile (and does not cost a fortune). And this option is not drugs, chiropractic, osteopathy, acupuncture, or any other SCAM – it is (physio)therapeutic exercise which is cheap, safe, and (mildly) effective.
During the coronavirus disease 2019 pandemic, Ayurvedic herbal supplements and homeopathic remedies were promoted as immune boosters (IBs) and disease-preventive agents. This happened in most parts of the world but nowhere more intensely than in India.
The present study examined the clinical outcomes among patients with chronic liver disease who presented with complications of portal hypertension or liver dysfunction temporally associated with the use of IBs in the absence of other competing causes. This Indian single-center retrospective observational cohort study included patients with chronic liver disease admitted for the evaluation and management of jaundice, ascites, or hepatic encephalopathy temporally associated with the consumption of IBs and followed up for 180 days. Chemical analysis was performed on the retrieved IBs.
From April 2020 to May 2021, 1022 patients with cirrhosis were screened, and 178 (19.8%) were found to have consumed complementary and alternative medicines. Nineteen patients with cirrhosis (10.7%), jaundice, ascites, hepatic encephalopathy, or their combination related to IBs use were included. The patients were predominantly male (89.5%). At admission, 14 (73.75%) patients had jaundice, 9 (47.4%) had ascites, 2 (10.5%) presented with acute kidney injury, and 1 (5.3%) had overt encephalopathy. Eight patients (42.1%) died at the end of the follow-up period. Hepatic necrosis and portal-based neutrophilic inflammation were the predominant features of liver biopsies.
Ten samples of IBs, including locally made ashwagandha powder, giloy juice, Indian gooseberry extracts, pure giloy tablets, multiherbal immune-boosting powder, other multiherbal tablets, and the homeopathic remedy, Arsenicum album 30C, were retrieved from our study patients. Samples were analyzed for potential hepatotoxic prescription drugs, known hepatotoxic adulterants, pesticides, and insecticides, which were not present in any of the samples. Detectable levels of arsenic (40%), lead (60%), and mercury (60%) were found in the samples analyzed. A host of other plant-derived compounds, industrial solvents, chemicals, and anticoagulants was identified using GC–MS/MS. These include glycosides, terpenoids, phytosteroids, and sterols, such as sitosterol, lupeol, trilinolein, hydroxy menthol, methoxyphenol, butyl alcohol, and coumaran derivatives.
The authors concluded that Ayurvedic and Homeopathic supplements sold as IBs potentially cause the worsening of preexisting liver disease. Responsible dissemination of scientifically validated, evidence-based medical health information from regulatory bodies and media may help ameliorate this modifiable liver health burden.
The authors comment that Ayurvedic herbal supplements and homeopathic remedies sold as IBs, potentially induce idiosyncratic liver injury in patients with preexisting liver disease. Using such untested advertised products can lead to the worsening of CLD in the form of liver failure or portal hypertension events, which are associated with a high risk of mortality compared to those with severe AH-related liver decompensation in the absence of timely liver transplantation. Severe mixed portal inflammation and varying levels of hepatic necrosis are common findings on liver histopathology in IB-related liver injury. Health regulatory authorities and print and visual media must ensure the dissemination of responsible and factual scientific evidence-based information on herbal and homeopathic “immune boosters” and health supplements to the public, specifically to the at-risk patient population.
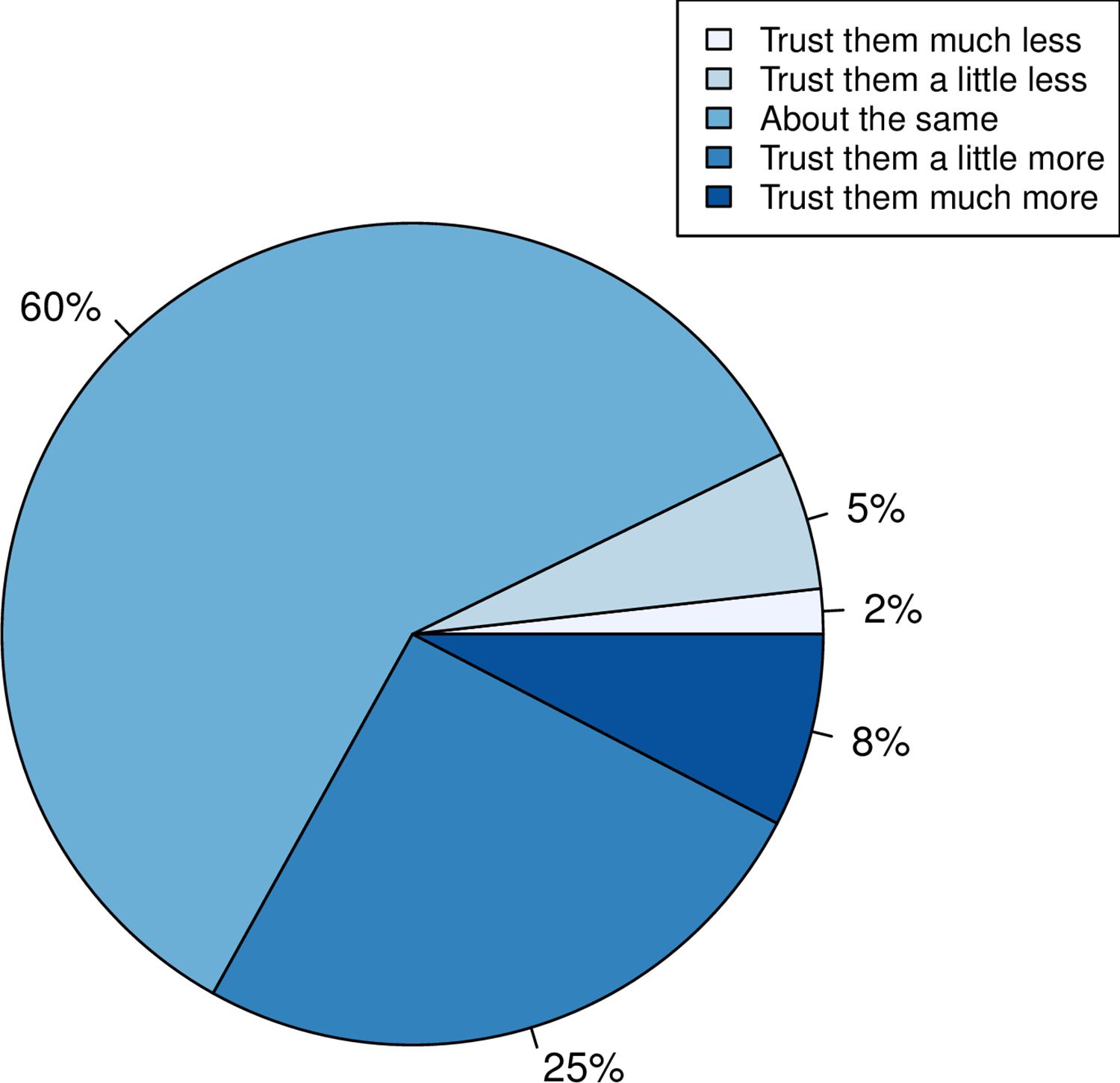
Research by the Milner Center for Evolution at the University of Bath, U.K., along with colleagues at the Universities of Oxford and Aberdeen, found that trust in scientists has hugely increased since the COVID-19 pandemic. The study also found that people were more likely to take the COVID-19 vaccine if their trust in the science had increased.
Using data from a survey of more than 2,000 U.K. adults commissioned by the Genetics Society, the team asked individuals whether their trust in scientists had gone up, down, or stayed the same.
- A third of people reported that their trust in scientists had gone up.
- When Pfizer, a company that made COVID-19 vaccines, was used as an example of the pharma industry, more people reported a positive response than when GlaxoSmithKline, a company not associated with the COVID-19 vaccine, was mentioned.
- The researchers also found that people who reported holding a negative view of science before the pandemic had become even more negative.
- People reporting increased trust were most likely to take the COVID-19 vaccine.
- Those preferring not to do so reported a decline in trust.
This is an interesting study with relevance to many discussions we had on this blog. I recommend reading it in full. Here are the abstract and link to the paper:
While attempts to promote acceptance of well-evidenced science have historically focused on increasing scientific knowledge, it is now thought that for acceptance of science, trust in, rather than simply knowledge of, science is foundational. Here we employ the COVID-19 pandemic as a natural experiment on trust modulation as it has enabled unprecedented exposure of science. We ask whether trust in science has on the average altered, whether trust has changed the same way for all and, if people have responded differently, what predicts these differences? We 1) categorize the nature of self-reported change in trust in “scientists” in a random sample of over 2000 UK adults after the introduction of the first COVID vaccines, 2) ask whether any reported change is likely to be real through consideration of both a negative control and through experiment, and 3) address what predicts change in trust considering sex, educational attainment, religiosity, political attitude, age and pre-pandemic reported trust. We find that many more (33%) report increased trust towards “scientists” than report decreased trust (7%), effects of this magnitude not being seen in negative controls. Only age and prior degree of trust predict change in trust, the older population increasing trust more. The prior degree of trust effect is such that those who say they did not trust science prior to the pandemic are more likely to report becoming less trusting, indicative of both trust polarization and a backfire effect. Since change in trust is predictive of willingness to have a COVID-19 vaccine, it is likely that these changes have public health consequences.
The Lightning Process (LP) is a therapy for ME based on ideas from osteopathy, life coaching, and neuro-linguistic programming. LP is claimed to work by teaching people to use their brains to “stimulate health-promoting neural pathways”. One young patient once described it as follows: “Whenever you get a negative thought, emotional symptom, you are supposed to turn on one side and with your arm movements in a kind if stop motion, just say STOP very firmly and that is supposed to cut off the adrenaline response.”
Allegedly, the LP teaches individuals to recognize when they are stimulating or triggering unhelpful physiological responses and to avoid these, using a set of standardized questions, new language patterns, and physical movements with the aim of improving a more appropriate response to situations. The LP involves three group sessions on consecutive days where participants are taught theories and skills, which are then practiced through simple steps, posture, and coaching.
Does LP work?
Some think it does, particularly in Norway, it seems.
Proponents of the ‘LP’ in Norway claim that 90% of all ME patients get better after trying it. However, such claims seem to be more than questionable.
- In the Norwegian ME association’s user survey from 2012 with 1,096 participants, 164 ME patients stated that they had tried LP. 21% of these patients experienced improvement or great improvement and 48% got worse or much worse.
- In Norway’s National Research Center in Complementary and Alternative Medicine, NAFKAM’s survey from 2015 amongst 76 patients 8 had a positive effect and 5 got worse or much worse.
- A survey by the Norwegian research foundation, published in the journal Psykologisk, with 660 participants, showed that 62 patients had tried LP, and 5 were very or fairly satisfied with the results.
Such figures seem to reflect the natural history of the condition and may be totally unrelated to LP.
The LP instructors’ claims of a 90% positive effect are used for marketing and for lobbying. Their aim is to influence politicians, health authorities, and welfare and disability benefits authorities. They want to get the LP course approved as part of the public health service.
The company ‘Aktiv Prosess’ was started by LP instructors Live Landmark and Vibeke C. Hammer. In an article in the Norwegian medical journal Tidsskriftet in 2016, Landmark describes her own customer satisfaction survey from 2008 as «generating a hypothesis». Landmark has also written a book about her personal story and holds lectures for medical students, medical doctors, and nurses. Now she is trying to run a clinical trial which, many experts believe, is far from rigorous and set up to produce a positive result.
Positive experiences with LP have received massive media coverage for 15 years. Anecdotes are recycled in the media and give the impression of being a higher number than reality. We rarely hear about those who deteriorated: https://lp-fortellinger.no/ (English language link here).
The NICE guidelines for ME/CFS specifically (and in my view rightly) warn against offering LP to ME patients.
I have been informed by the publisher, that my book has been published yesterday. This is about two months earlier than it was announced on Amazon. It is in German – yes, I have started writing in German again. But not to worry, I translated the preface for you:
Anyone who falls ill in Germany and therefore needs professional assistance has the choice, either to consult a doctor or a non-medical practitioner (Heilpraktiker).
– The doctor has studied and is licensed to practice medicine; the Heilpraktiker is state-recognized and has passed an official medical examination.
– The doctor is usually in a hurry, while the Heilpraktiker takes his time and empathizes with his patient.
– The doctor usually prescribes a drug burdened with side effects, while the Heilpraktiker prefers the gentle methods of alternative medicine.
So who should the sick person turn to? Heilpraktiker or doctor? Many people are confused by the existence of these parallel medical worlds. Quite a few finally decide in favor of the supposedly natural, empathetic, time-tested medicine of the Heilpraktiker. The state recognition gives them the necessary confidence to be in good hands there. The far-reaching freedoms the Heilpraktiker has by law, as well as the coverage of costs by many health insurances, are conducive to further strengthening this trust. “We Heilpraktiker are recognized and respected in politics and society,” writes Elvira Bierbach self-confidently, the publisher of a standard textbook for Heilpraktiker.
The first consultation of our model patient with the Heilpraktiker of his choice is promising. The Heilpraktiker responds to the patient with understanding, usually takes a whole hour for the initial consultation, gives explanations that seem plausible, is determined to get to the root of the problem, promises to stimulate the patient’s self-healing powers naturally, and invokes a colossal body of experience. It almost seems as if our patient’s decision to consult a Heilpraktiker was correct.
However, I have quite significant reservations about this. Heilpraktiker are perhaps recognized in politics and society, but from a medical, scientific, or ethical perspective, they are highly problematic. In this book, I will show in detail and with facts why.
The claim of government recognition undoubtedly gives the appearance that Heilpraktiker are adequately trained and medically competent. In reality, there is no regulated training, and the competence is not high. The official medical examination, which all Heilpraktiker must pass is nothing more than a test to ensure that there is no danger to the general public. The ideas of many Heilpraktiker regarding the function of the human body are often in stark contradiction with the known facts. The majority of Heilpraktiker-typical diagnostics is pure nonsense. The conditions that they diagnose are often based on little more than naive wishful thinking. The treatments that Heilpraktiker use are either disproven or not proven to be effective.
There is no question in my mind that Heilpraktiker are a danger to anyone who is seriously ill. And even if Heilpraktiker do not cause obvious harm, they almost never offer what is optimally possible. In my opinion, patients have the right to receive the most effective treatment for their condition. Consumers should not be misled about health-related issues. Only those who are well-informed will make the right decisions about their health.
My book provides this information in plain language and without mincing words. It is intended to save you from a dangerous misconception of the Heilpraktiker profession. Medical parallel worlds with the radically divergent quality standard – doctor/Heilpraktiker – are not in the interest of the patient and are simply unacceptable for an enlightened society.
The ‘keto diet’ is a currently popular high-fat, low-carbohydrate diet; it limits the intake of glucose which results in the production of ketones by the liver and their uptake as an alternative energy source by the brain. It is said to be an effective treatment for intractable epilepsy. In addition, it is being promoted as a so-called alternative medicine (SCAM) for a wide range of conditions, including:
- weight loss,
- cognitive and memory enhancement,
- type II diabetes,
- cancer,
- neurological and psychiatric disorders.
Now, it has been reported that the ‘keto diet’ may be linked to higher levels of cholesterol and double the risk of cardiovascular events. In the study, researchers defined a low-carb, high-fat (LCHF) diet as 45% of total daily calories coming from fat and 25% coming from carbohydrates. The study, which has so far not been peer-reviewed, was presented Sunday at the American College of Cardiology’s Annual Scientific Session Together With the World Congress of Cardiology.
“Our study rationale came from the fact that we would see patients in our cardiovascular prevention clinic with severe hypercholesterolemia following this diet,” said Dr. Iulia Iatan from the Healthy Heart Program Prevention Clinic, St. Paul’s Hospital, and University of British Columbia’s Centre for Heart Lung Innovation in Vancouver, Canada, during a presentation at the session. “This led us to wonder about the relationship between these low-carb, high-fat diets, lipid levels, and cardiovascular disease. And so, despite this, there’s limited data on this relationship.”
The researchers compared the diets of 305 people eating an LCHF diet with about 1,200 people eating a standard diet, using health information from the United Kingdom database UK Biobank, which followed people for at least a decade. They found that people on the LCHF diet had higher levels of low-density lipoprotein and apolipoprotein B. Apolipoprotein B is a protein that coats LDL cholesterol proteins and can predict heart disease better than elevated levels of LDL cholesterol can. The researchers also noticed that the LCHF diet participants’ total fat intake was higher in saturated fat and had double the consumption of animal sources (33%) compared to those in the control group (16%). “After an average of 11.8 years of follow-up – and after adjustment for other risk factors for heart disease, such as diabetes, high blood pressure, obesity, and smoking – people on an LCHF diet had more than two times higher risk of having several major cardiovascular events, such as blockages in the arteries that needed to be opened with stenting procedures, heart attack, stroke, and peripheral arterial disease.” Their press release also cautioned that their study “can only show an association between the diet and an increased risk for major cardiac events, not a causal relationship,” because it was an observational study, but their findings are worth further investigation, “especially when approximately 1 in 5 Americans report being on a low-carb, keto-like or full keto diet.”
I have to say that I find these findings not in the slightest bit surprising and would fully expect the relationship to be causal. The current craze for this diet is concerning and we need to warn consumers that they might be doing themselves considerable harm.
Other authors have recently pointed out that, within the first 6-12 months of initiating the keto diet, transient decreases in blood pressure, triglycerides, and glycosylated hemoglobin, as well as increases in HDL and weight loss may be observed. However, the aforementioned effects are generally not seen after 12 months of therapy. Despite the diet’s favorable effect on HDL-C, the concomitant increases in LDL-C and very-low-density lipoproteins (VLDL) may lead to increased cardiovascular risks. And another team of researchers has warned that “given often-temporary improvements, unfavorable effects on dietary intake, and inadequate data demonstrating long-term safety, for most individuals, the risks of ketogenic diets may outweigh the benefits.”
Norbert Hofer is the former leader of the Austrian right-wing FPÖ party who almost became Austria’s President. Currently, he is the 3rd member of the National Council. Hofer is a man full of surprises; he stated, for instance, that the Quran was more dangerous than COVID-19 during a speech held at a 2020 campaign event. As a result, he was sued for hate-speech.
Hofer’s latest coup is not political but commercial: Hofer is launching his own dietary supplement on the market. It is called “Formula Fortuna” and contains:
- L-tryptophan; a Cochrane review concluded that “a large number of studies appear to address the research questions, but few are of sufficient quality to be reliable. Available evidence does suggest these substances are better than placebo at alleviating depression. Further studies are needed to evaluate the efficacy and safety of 5‐HTP and tryptophan before their widespread use can be recommended. The possible association between these substances and the potentially fatal Eosinophilia‐Myalgia Syndrome has not been elucidated. Because alternative antidepressants exist which have been proven to be effective and safe the clinical usefulness of 5‐HTP and tryptophan is limited at present.”
- Hydroxypropylmethylcellulose, a common delivery system.
- Rhodiola rosea extracts; human studies evaluating R. rosea did not have sufficient quality to determine whether it has properties affecting fatigue or any other condition.The U.S. Food and Drug Administration (FDA) has issued warning letters to manufacturers of R. rosea dietary supplement products unapproved as new drugs, adulterated, misbranded and in federal violation for not having proof of safety or efficacy for the advertised conditions of alleviating Raynaud syndrome, altitude sickness, depression or cancer.
- Ginseng root extract. Although ginseng has been used in traditional medicine for centuries, modern research is inconclusive about its biological effects. Preliminary clinical research indicates possible effects on memory, fatigue, menopause symptoms, and insulin response in people with mild diabetes. Out of 44 studies examined between 2005–2015, 29 showed positive, limited evidence, and 15 showed no effects. As of 2021, there is insufficient evidence to indicate that ginseng has any health effects. A 2021 review indicated that ginseng had “only trivial effects on erectile function or satisfaction with intercourse compared to placebo”. The constituents include steroid saponins known as ginsenosides, but the effects of these ginseng compounds have not been studied with high-quality clinical research as of 2021, and therefore remain unknown. As of 2019, the United States FDA and Federal Trade Commission have issued numerous warning letters to manufacturers of ginseng dietary supplements for making false claims of health or anti-disease benefits, stating that the “products are not generally recognized as safe and effective for the referenced uses” and are illegal as unauthorized “new drugs” under federal law. Concerns exist when ginseng is used chronically, potentially causing side effects such as headaches, insomnia, and digestive problems. Ginseng may have adverse effects when used with the blood thinner warfarin. Ginseng also has adverse drug reactions with phenelzine, and a potential interaction has been reported with imatinib, resulting in hepatotoxicity, and with lamotrigine. Other side effects may include anxiety, insomnia, fluctuations in blood pressure, breast pain, vaginal bleeding, nausea, or diarrhea.
- Zinc gluconate which has been used in lozenges for treating the common cold. However, controlled trials with lozenges which include zinc acetate have found it has the greatest effect on the duration of coldsInstances of anosmia (loss of smell) have been reported with intranasal use of some products containing zinc gluconate. In September 2003, Zicam faced lawsuits from users who claimed that the product, a nasal gel containing zinc gluconate and several inactive ingredients, negatively affected their sense of smell and sometimes taste. Some plaintiffs alleged experiencing a strong and very painful burning sensation when they used the product. Matrixx Initiatives, Inc., the maker of Zicam, responded that only a small number of people had experienced problems and that anosmia can be caused by the common cold itself. In January 2006, 340 lawsuits were settled for $12 million.
- Pyridoxine hydrochloride (vitamin B6) is usually well tolerated, though overdose toxicity is possible. Occasionally side effects include headache, numbness, and sleepiness. Pyridoxine overdose can cause a peripheral sensory neuropathy characterized by poor coordination, numbness, and decreased sensation to touch, temperature, and vibration.
‘Formula Fortuna’ allegedly is for lifting your mood. If I, however, tell you that you need to pay one Euro per day for the supplement, your mood might even change in the opposite direction.
What next?
I think I might design a dietary supplement against stupidity. It will not carry any of the risks of Hofer’s new invention but, I am afraid, it might be just as ineffective as Hofer’s ‘Formual Fortuna’.
A recent article in ‘The Lancet Regional Health‘ emphasized the “need for reimagining India’s health system and the importance of an inclusive approach for Universal Health Coverage” by employing traditional medicine, including homeopathy. This prompted a response by Siddhesh Zadey that I consider worthy of reproducing here in abbreviated form:
… Since the first trial conducted in 1835 that questioned homeopathy’s efficacy, multiple randomized controlled trials (RCTs) and other studies compiled in several systematic reviews and meta-analyses have shown that there is no reliable and clinically significant effect of non-individualized or individualized homeopathic treatments across disease conditions ranging from irritable bowel syndrome in adults to acute respiratory tract infections in children when compared to placebo or other treatments. Even reviews that support homeopathy’s efficacy consistently caution about low quality of evidence and raise questions on its clinical use. The most recent analysis of reporting bias in homeopathic trials depicted problematic trial conduction practices that further obscure reliability and validity of evidence. Homeopathic treatments have also been linked to aggravations and non-fatal and fatal adverse events.
The Lancet has previously published on another kind of harm that uptake of homeopathy encourages in India: delay to evidence-based clinical care that can lead to fatality. Authors have pointed out that evidence for some of the alternative systems of medicine may not come from RCTs. I agree that more appropriate study designs and analytical techniques are needed for carefully studying individualized treatment paradigms. However, the need for agreement on some consistent form of evidence synthesis and empirical testing across diverse disciplines cannot be discounted. Several other disciplines including psychology, economics, community health, implementation science, and public policy have adopted RCTs and related study designs and have passed the empirical tests of efficacy. Moreover, the ideas around mechanism of action in case of homeopathy still remain controversial and lack evidence after over a century. On the contrary, biochemical, molecular, and physiological mechanistic evidence supporting allopathic treatments has grown abundantly in the same period.
Owing to lack of evidence on its efficacy and safety, the World Health Organization had previously warned against the use of homeopathic treatments for severe diseases. Additionally, multiple countries, including Germany where the practice originated, have initiated mechanisms that discourage uptake of homeopathy while others are considering banning it. Homeopathy doesn’t work, could be harmful, and is not a part of Indian traditional medicine. While we should welcome pluralistic approaches towards UHC, we need to drop homeopathy.
(for references, see original text)
___________________
Yes, in the name of progress and in the interest of patients, “we need to drop homeopathy” (not just in India but everywhere). I quite agree!