A recent article about ayurvedic medicine caught my eye. Here are a few excerpts:
Imagine if there were a magic pill to ward off COVID-19. Or if you could cure diabetes with vegetable juices and herbal pills instead of controlling it with insulin medication. Or if yoga and breathing exercises were all you need to do to get rid of asthma. These are all claims made by Patanjali Ayurved, one of India’s biggest manufacturers of traditional ayurvedic products…
Many scientists have expressed concerns over the lack of research into the safety and efficacy of ayurvedic products… Nonetheless, Ayurveda enjoys widespread acceptance among Indians. And under India’s Hindu-nationalist government that took power in 2014, ayurveda and other alternative systems of medicine have received unprecedented government support. India’s ministry of alternative medicine gets nearly $500 million a year. The government also promotes ayurveda through its international trade and diplomatic channels. All this set Patanjali’s fortunes soaring.
But now the Supreme Court of India has temporarily banned Patanjali – named after a Hindu mystic best known for his writings on yoga – from advertising some of its products… “The entire country has been taken for a ride,” Ahsanuddin Amanullah, one of the two judges conducting the court hearing, told the lawyer representing the government… The Indian Medical Association had brought the case to court in August 2022, claiming that Patanjali and its brand ambassador Baba Ramdev made a series of false claims against evidence-backed modern medicine and its practitioners, and spread misinformation about COVID-19 vaccines. Their petition also referred to instances where Ramdev lambasted modern medicine as a “stupid and bankrupt science” at a yoga session. The trigger was a series of Patanjali advertisements in Indian newspapers in July 2022 claiming that ayurvedic products could cure chronic conditions like diabetes, high blood pressure, heart diseases and autoimmune conditions. The Indian Medical Association’s petition alleged that such claims were in violation of India’s Drugs and Magic Remedies (Objectionable Advertisements) Act.
…The company’s public face – yoga guru Baba Ramdev – is a vocal supporter of India’s ruling party, the BJP, and Prime Minister Narendra Modi. Modi even inaugurated Patanjali’s ayurvedic research facility in 2017… Some scientists have accused their government of promoting these alternative medicines at the expense of modern medicine, partly as a way to glorify India’s culture and history. “One of the political ideas of this government is to glorify the Hindu tradition,” says Dhrubajyoti Mukherjee, president of the Breakthrough Science Society, an organization that promotes scientific thinking. “But in the name of our glorious past, the government is propagating obscure, unscientific ideas.”
… A few months after the outbreak of the COVID-19 pandemic in 2020, India’s health minister at the time, Harsh Vardhan participated in the company’s launch of pills, where Ramdev, the yoga guru, claimed the pills showed “100 percent favorable results” during clinical trials on patients. Despite experts flagging the lack of evidence, the company said it sold 2.5 million kits in six months, consisting of the tablets to ward off COVID-19 and bottled oils that would allegedly boost immunity. And the company is making an enormous amount of money: Its income was over $1.3 billion in the financial year 2021-22, with profits of $74 million before taxes.
Addressing the overall impact of misinformation about ayurvedic treatments, Dr. Jayesh Lele, vice president of the Indian Medical Association, says “Our worry is people are being misguided. We have got people who’ve left our treatment saying their kidneys will be able to function properly [using ayurvedic medicines] and ended up with renal failure. The same happened with patients suffering from hepatitis, who’ve got the wrong medicine and ended up with further problems. And if you say every day that modern medicine is bad, that is not acceptable.”
_______________
The sad thing, in my view, is that (as discussed previously) ayurvedic medicine has not just taken India for a ride:
- King Charles’ “Ayurvedic Centre of Excellence” turns out to be an embarrassing failure
- Dr Michael Dixon seems to support homeopathy as a treatment for cancer
- PRINCE CHARLES: the ‘immense value’ of alternative diagnostic methods
- Will the UK ‘ROYAL COLLEGE OF GENERAL PRACTITIONERS’ soon become a ‘ROYAL COLLEGE OF QUACKERY’?
And perhaps even more disappointing is the notion that, while in India they take action in order to prevent harm, I can see no such developments in the UK.
The origin of coronavirus 2 (SARS-CoV-2) has been the subject of intense speculation and several conspiracy theories, not least amongst the enthusiasts of so-called alternative medicine. Now Australian scientists have attempted to identify the origin of the coronavirus 2 (SARS-CoV-2). As this is undoubtedly a most sensitive subject, let me show you the unadulterated abstract of their paper:
The origin of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is contentious. Most studies have focused on a zoonotic origin, but definitive evidence such as an intermediary animal host is lacking. We used an established risk analysis tool for differentiating natural and unnatural epidemics, the modified Grunow–Finke assessment tool (mGFT) to study the origin of SARS-COV-2. The mGFT scores 11 criteria to provide a likelihood of natural or unnatural origin. Using published literature and publicly available sources of information, we applied the mGFT to the origin of SARS-CoV-2. The mGFT scored 41/60 points (68%), with high inter-rater reliability (100%), indicating a greater likelihood of an unnatural than natural origin of SARS-CoV-2. This risk assessment cannot prove the origin of SARS-CoV-2 but shows that the possibility of a laboratory origin cannot be easily dismissed.
The somewhat clumsy wording harbours explosive potential. It is more likely that the pandemic was started by a laboratory accident than by a zoonosis. In this case, it would be man-made rather than natural. The authors of the paper do, however, caution that their analysis does not prove the origin of the coronavirus. They merely speak of likelihoods. Moreover, it seems important to stress that there is no scientific evidence that Sars-CoV-2 was deliberately developed as a biological warfare agent.
Will this paper put an end to speculation and conspiracy?
I doubt it!
The Amercian Medical Association (AMA) recently published a lengthy article on naturopathy in the US. Here are some excerpts:
There are three types of health professionals who offer naturopathic treatment:
- Naturopathic doctors. These nonphysicians graduate from a four-year, professional-level program at an accredited naturopathic medical school, earning either the doctor of naturopathy (ND) degree or the doctor of naturopathic medicine (NMD) degree.
- Traditional naturopaths, who have obtained education through some combination of a mentorship program with another professional or at an alternative clinic, distance-learning program or classroom schooling on natural health, or other holistic studies.
- Other health professionals such as chiropractors, massage therapists, dentists, nurses, nutritionists, or physicians who practice under a professional license but include some naturopathic methods in their practice and who may have studied on their own or taken courses on naturopathic methods.
At least 24 states and the District of Columbia regulate the practice of naturopathy. In order to be licensed, naturopaths in these states must earn an ND or NMD from an accredited naturopathic program and pass the Naturopathic Physicians Licensing Exam. Three states—Florida, South Carolina and Tennessee—prohibit the practice of naturopathy. In states that neither license nor prohibit the practice of naturopathy, traditional naturopaths and NDs alike may practice without being subject to state regulation.
Postgraduate training is neither common nor required of graduates of naturopathic schools, except in Utah … less than 10% of naturopaths participate in an approved residency, and such residencies last only a year and lack a high degree of standardization.
… naturopaths are required to get at least 1,200 hours of direct patient contact, physicians get 12,000–16,000 hours of clinical training…
ND programs emphasize naturopathic principes—for example, the healing power of nature—and naturopathic therapeutics such as botanical medicine, homeopoathy and hydrotherapy. Coursework in naturopathic therapeutics is combined with, and taught alongside, coursework in sciences. But there are no specifications around the number of hours required in each area … naturopathic students may lack exposure to key clinical scenarios in the course of their training … naturopathic students’ clinical experience is typically gained through outpatient health care clinics, as naturopathic medical schools typically do not have significant hospital affiliation. This means there is no guarantee that a naturopathic student completing a clinical rotation will see patients who are actually sick or hospitalized, and they may not be exposed to infants, children, adolescents or the elderly. It has been said that naturopaths tend to treat the “worried well.”
… Naturopaths claim they are trained as primary care providers and, as such, are educated and trained to diagnose, manage and treat many conditions, including bloodstream infections, heart disease and autoimmune disorders. Yet their education and training falls several years and thousands of hours short of what physicians get.
…The AMA believes it is the responsibility of policymakers to ensure that naturopaths’ claims that they can treat a broad range of conditions are backed by facts—facts that include the specific education and training necessary to ensure patient safety.
________________
The AMA is clearly cautious here. A less polite statement might simply stress that naturopaths are taught a lot of nonsense which they later tend to administer to their unsuspecting patients. On this blog, we have repeatedly discussed the danger naturopaths present to public health in the US and elsewhere, e.g.:
- How reliable are the claims made by naturopathic influencers?
- Naturopath jailed for selling fraudulent vaccination documents
- Naturopath fined for misdiagnosing and treating a rectal tumor for hemorrhoids
- Naturopaths are ‘not bound by science,’ lawyer argues
- Vaccination rates of Canadian healthcare professionals: those of chiropractors and naturopaths are at the lowest
- Is veterinary naturopathy animal abuse?
- Naturopathic ‘cancer specialist’ using coffee enemas found guilty
- Patients consulting chiropractors, homeopaths, or naturopaths are less likely to agree to the flu jab
- A naturopath responsible for the death of two cancer patients was sentenced to two years
- A naturopath in court after two of his cancer patients died
- Many naturopaths, homeopaths, and chiropractors are a risk to public health
- Naturopath treats autism with fecal transplants
- A naturopath promoting fake news about COVID vaccinations
- Naturopathy (according to the WNF) = quackery steeped in obsolete fantasies
- Canadian naturopaths may no longer call themselves ‘medically trained’
- Naturopaths’ counselling against vaccinations could be criminally negligent
- Naturopathy for cancer … claims that have the potential to be lethal
- Severe liver injury due to naturopaths’ prescription of Epsom salt
- Naturopaths should not treat children
- Some naturopaths are clearly a danger to public health
- Death of a child through naturopathy
Claims that naturopaths are a viable alternative to evidence-based medicine are wrong, irresponsible and dangerous. Regulators must be reminded that they have the duty to protect the public from charlatans and should therefore ensure that no false therapeutic or diagnostic claims can be made by naturopaths.
Please allow me to write a post today that has nothing at all to do with so-called alternative medicine, not even with medicine.
Yesterday, 13 March, was my mother’s birthday. She was born in 1911 and died in 1989. I often think of her and the remarkable life she had, particularly, of course, on 13 March.
Going through my X-feed yesterday, I saw this post from the Auschwitz Memorial:
Józef Kowalski was born in Siedliska on 13 March 1911 to Wojciech and Zofia Borowiec, the seventh of their nine children. He was ordained a priest on 29 May 1938 as member of the Salesian Religious Society. During the German occupation of Poland, the Salesians continued their educational work. The Gestapo arrested Kowalski on 23 May 1941, along with eleven other Salesians. They were taken to Montelupich Prison and tortured.
Kowalski was sent to Auschwitz on 26 June 1941. There, he ministered to his fellow prisoners usually in secret, but at least once in front of everyone at the moment of mass execution. He was ordered by Blockführer Gerhard Palitzsch to trample upon his rosary but refused. Palitzsch was one of the most brutal of all the SS officers of the camps (he later vanished from the records, possibly killed in action). As punishment, he was assigned to a penal company. In his last letter to his parents, Józef wrote:
Do not worry about me; I am in God’s hands. I want to assure you that I feel His help at every step. Despite the present situation, I am happy and completely at peace.
On 3 July 1942 he was mocked and severely beaten by the guards for being a priest. The same night, they pulled him out of his barracks, gravely beat him again and killed him. Kowalski’s body was found the next day and burned with others. He was 31 years of age.
Pope John Paul II who had known Kowalski personally beatified Kowalski on 13 June 1999.
____________________________
Please forgive me if you find this post irrelevant or inappropriate.
Looking at some ancient papers of mine, I came across a short BMJ paper from 1994. Here is a passage from it:
… A standard letter (on departmental letterhead) was written (in German) to all 189 firms that we identified as marketing herbal drugs in Germany. It asked (among other questions) for reprints of articles reporting controlled clinical trials on the company’s product(s).
Only 19 replies had reached us six weeks later. Four of these included at least one reprint. Twelve respondents regretted not knowing of clinical trials on their drug(s). In three cases we had written to a wrong address (one
instance) or to a firm which did not market phytomedicines (two instances).
These data, though far from conclusive, do not give the impression that research is in proportion to either prevalence or financial tumover of herbal remedies…
I wonder what the results would be, if we repeated this little excercise today, 30 years afteer the original investigation. I fear that the findings would be much the same or perhaps even worse. I also suspect that they would be similar regardless of the country we chose. Those who sell herbal remedies have very little incentive to do expensive clinical trials to test whether the products they earn their money with actually work. They may be doing well without it and ask themselves, why spend money on research that might not show what we hope and could easily turn out to jeopardize our financial success?
But the problem is by no means confined to herbal manufacturers (who would arguably have an important share to initiate and sponsor research). Even though fundamental questions remain unanswered, research into herbal medicine is scarce across the board.
To see whether this statement is true, I did a very quick Medline search. It showed that, in 2023, just over 13 000 papers on herbal medicine emerged. Of those, just 460 were listed as clinical trials. The latter figure is almost certainly considerably smaller than the true amount because Medline is over-generous in classifying papers as clinical trials. I thus estimate that only around 200 clinical trials of herbal medicine are conducted each year. Considering that we are dealing with thousands of herbs and ten thousands of herbal products, this figure is an embarrassment for the sector – which, as we have seen just days ago, is doing extremely well in finacial terms.
Dry needling is a therapy that is akin to acupuncture and trigger point therapy. It is claimed to be safe – but is this true?
Researchers from Ghent presented a series of 4 women aged 28 to 35 who were seen at the emergency department (ED) with post-dry needling pneumothorax between September 2022 and December 2023. None of the patients had any relevant medical history. All had been treated for a painful left shoulder, trapezius muscle or neck region in outpatient physiotherapist practices. At least three different physiotherapists were involved.
One patient presented to the ER on the same day as the dry needling procedure, the others presented the day after. All mentioned thoracic pain and dyspnoea. Clinical examination in all of these patients was unremarkable, as were their vital signs. Diagnosis was confirmed with ultrasound (US) and chest X-ray (CXR) in all patients. The latter exam showed left-sided apical pleural detachment with a median of 3.65 cm in expiration.
Two patients were managed conservatively. One patient (initial pneumothorax 2.5 cm) was discharged. The US two days later displayed a normally expanded lung. One patient with an initial apical size of 2.8 cm was admitted with 2 litres of oxygen through a nasal canula and discharged from the hospital the next day after US had shown no increase in size. Her control CXR 4 days later showed only minimal pleural detachment measuring 6 mm. The two other patients were treated with US guided needle aspiration. One patient with detachment initially being 4.5 cm showed decreased size of the pneumothorax immediately after aspiration. She was admitted to the respiratory medicine ward and discharged the next day. Control US and CXR after 1 week showed no more signs of pneumothorax. In the other patient, with detachment initially being 5.5 cm, needle aspiration resulted in complete deployment on US immediately after the procedure, but control CXR showed a totally collapsed lung 3 hours later. A small bore chest drain was placed but persistent air leakage was seen. Several trials of clamping the drain resulted in recurrent collapsing of the lung. After CT-scan had shown no structural deformities of the lung, suction was gradually reduced and the drain was successfully removed on the sixth day after placement. The patient was then discharged home. Control CXR 3 weeks later was normal.
The authors concluded that post-dry needling pneumothorax is, contrary to numbers cited in literature, not extremely rare. With rising popularity of the technique we expect complications to occur more often. Patients and referring doctors should be aware of this. In their informed consent practitioners should mention pneumothorax as a considerable risk of dry needling procedures in the neck, shoulder or chest region.
The crucial question, in my view, is this: do the risks of dry-needling out weigh the risks of this form of therapy? Let’s have a look at some of the recent evidence that we discussed on this blog:
- Spinal Manipulation and Electrical Dry Needling for Subacromial Pain Syndrome: A Nonsensical Trial
- Dry needling is useless for rehabilitation after shoulder surgery
- High velocity, low amplitude techniques are not superior to no treatment in the management of tension-type headache
- Which treatments are best for acute and subacute mechanical non-specific low back pain? A systematic review with network meta-analysis
- Acupuncture for chronic pain: the new NICE guideline
- Acupuncture for the Relief of Chronic Pain? A new, thorough synthesis fails to produce strong evidence that acupuncture works
The evidence is clearly mixed and unconvincing. I am not sure whether it is strong enough to afford a positive risk/benefit balance. In other words: dry needling is a therapy that might best be avoided.
I usually take ‘market reports’ with a pinch of salt. Having said that, this document makes some rather interesting predictions:
The size of the market for so-called alternative medicine (SCAM) is projected to expand from USD 147.7 billion in 2023 to approximately USD 1489.4 billion by the year 2033. This projection indicates a remarkable Compound Annual Growth Rate (CAGR) of 26% over the forecast period.
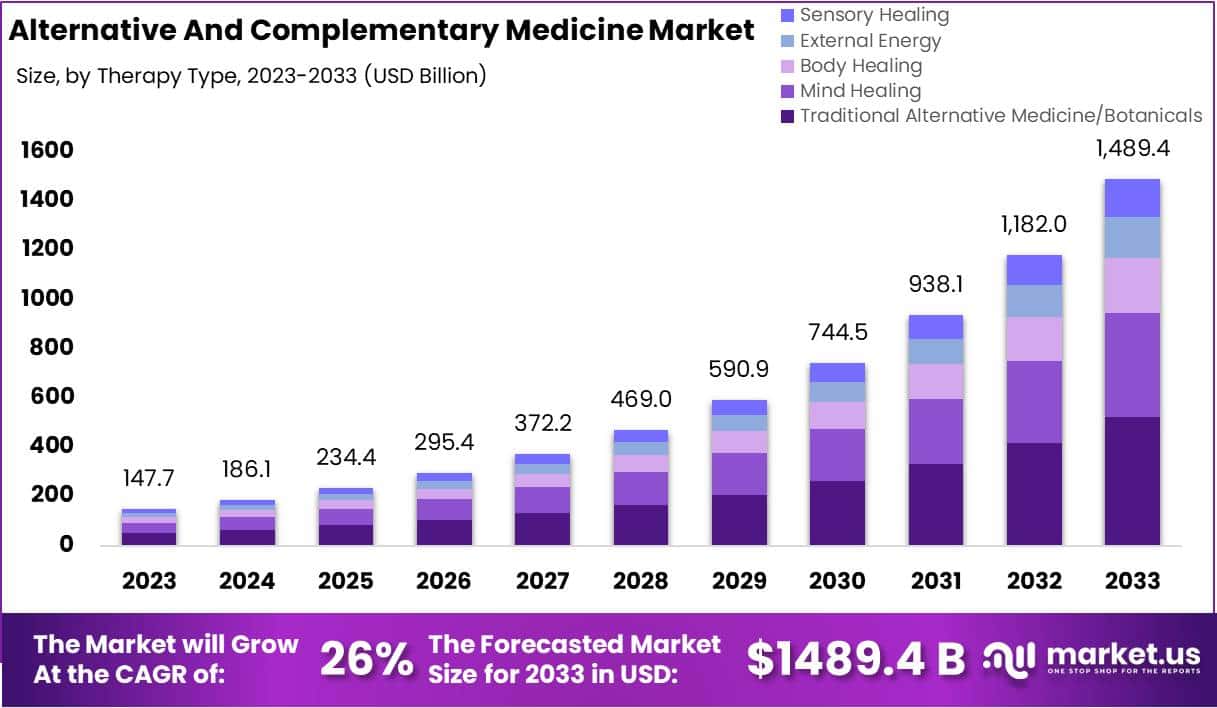
The market for SCAM is experiencing significant growth, fueled by increasing consumer interest in natural and holistic health solutions. This trend reflects a broader shift in societal attitudes towards health and wellness, emphasizing preventive care and natural health practices.
The market’s dynamics are influenced by various factors, including consumer preferences, regulatory standards, and evolving perceptions of health and wellness. As the popularity of these alternative therapies grows, it is crucial for individuals to consult with healthcare professionals to ensure that these non-conventional approaches are safely and effectively incorporated into their overall health regimen. The increasing acceptance of SCAM underscores a collective move towards more personalized and holistic healthcare solutions, resonating with today’s health-conscious consumers.
In 2023, Traditional Alternative Medicine/Botanicals led the market, capturing a 35.2% share, which reflects a strong consumer inclination towards these treatments. Dietary Supplements were prominent in the market, securing a 25.1% share in 2023, which underscores the high consumer demand for nutritional aids. Direct Sales were the most favored distribution channel, accounting for 43.2% of the market share in 2023, which indicates their significant impact on guiding consumer purchases. Pain Management was the predominant application area, holding a 24.9% market share in 2023, propelled by the growing acknowledgment of non-pharmacological treatment options. Adults represented a substantial portion of the market, making up 62.33% in 2023, signifying a marked preference for SCAM therapies within this age group. Europe stood out as the market leader, claiming a 42.6% share in 2023, a position supported by widespread acceptance, governmental backing, and an increasing elderly population. The regions of North America and Asia-Pacific are highlighted as areas with potential, signaling opportunities for market expansion beyond the European stronghold in the upcoming years.
Leading Market Players Are:
- Columbia Nutritional
- Nordic Nutraceuticals
- Ramamani Iyengar Memorial Yoga Institute
- The Healing Company Ltd.
- John Schumacher Unity Woods Yoga Centre
- Sheng Chang Pharmaceutical Company
- Pure encapsulations LLC.
- Herb Pharm
- AYUSH Ayurvedic Pte Ltd.
Recent developments:
- In December 2023, Adoratherapy launched the Alkemie Chakra Healing Line, an aromatherapy range aimed at harmonizing the seven chakras.
- Coworth Park introduced the Hebridean Sound Treatment in October 2023, merging traditional Hebridean sounds with guided meditation to offer a novel, restorative wellness experience.
- The World Health Organization released draft guidelines in September 2023 for the safe, effective application of traditional medicines.
- Telehealth services, expanding significantly in August 2023, have broadened the reach of SCAM, enhancing patient access to these treatments.
Microplastics are tiny polymer fragments that range from less than 0.2 inch to 1/25,000th of an inch. Smaller particles are called nanoplastics and are measured in billionths of a metre. Microplastics and nanoplastics (MNPs) are emerging as a potential risk factor for human health and for cardiovascular disease in particular. However, direct evidence that this risk extends to humans has so far been lacking. This investigation is a first step towards filling the gap.
The researchers conducted a prospective, multicenter, observational study involving patients who were undergoing carotid endarterectomy for asymptomatic carotid artery disease. The excised carotid plaque specimens were analyzed for the presence of MNPs with the use of pyrolysis–gas chromatography–mass spectrometry, stable isotope analysis, and electron microscopy. Inflammatory biomarkers were assessed with enzyme-linked immunosorbent assay and immunohistochemical assay. The primary end point was a composite of myocardial infarction, stroke, or death from any cause among patients who had evidence of MNPs in plaque as compared with patients with plaque that showed no evidence of MNPs.
A total of 304 patients were enrolled in the study, and 257 completed a mean (±SD) follow-up of 33.7±6.9 months. Polyethylene was detected in carotid artery plaque of 150 patients (58.4%), with a mean level of 21.7±24.5 μg per milligram of plaque; 31 patients (12.1%) also had measurable amounts of polyvinyl chloride, with a mean level of 5.2±2.4 μg per milligram of plaque. Electron microscopy revealed visible, jagged-edged foreign particles among plaque macrophages and scattered in the external debris. Radiographic examination showed that some of these particles included chlorine. Patients in whom MNPs were detected within the atheroma were at higher risk for a primary end-point event than those in whom these substances were not detected (hazard ratio, 4.53; 95% confidence interval, 2.00 to 10.27; P<0.001).
The authors concluded that, in this study, patients with carotid artery plaque in which MNPs were detected had a higher risk of a composite of myocardial infarction, stroke, or death from any cause at 34 months of follow-up than those in whom MNPs were not detected.
This is an impressive study – so much so that I report it here even though it has no connection to so-called alternative medicine, the focus of my blog. The fact that 58% of all plaques contained MNPs seems alarming. The finding that the presence of these MNPs is associated with a poor cardiovascular prognosis seems even more concerning.
MNPs have been found in every environmental compartment on earth. They are ingested not just by humans but by most animals as well. Even though research into these issues is most active, their effects are so far still under-researched and not fully understood.
The authors of the new investigation are rightly cautious: “Our data must be confirmed by other studies and on larger populations,” said Marfella, professor of internal medicine and director of the department of medical and surgical sciences at the University of Campania Luigi Vanvitelli in Naples, Italy. “However, our study convincingly highlights the presence of plastics and their association with cardiovascular events in a representative population affected by atherosclerosis.”
Of course, many questions are as yet unanswered but the subject is as worrying as it is important, e.g.:
- Should exposure to MNPs be considered a cardiovascular risk factor?
- What organs in addition to the heart may be at risk?
- How can we reduce exposure?”
I wish I knew the ansers.
According to its authors, this study‘s objective was to demonstrate that acupuncture is beneficial for decreasing the risk of ischaemic stroke in patients with rheumatoid arthritis (RA).
The investigation was designed as a propensity score-matched cohort nationwide population-based study. Patients with RA diagnosed between 1 January 1997 and 31 December 2010, through the National Health Insurance Research Database in Taiwan. Patients who were administered acupuncture therapy from the initial date of RA diagnosis to 31 December 2010 were included in the acupuncture cohort. Patients who did not receive acupuncture treatment during the same time interval constituted the no-acupuncture cohort. A Cox regression model was used to adjust for age, sex, comorbidities, and types of drugs used. The researchers compared the subhazard ratios (SHRs) of ischaemic stroke between these two cohorts through competing-risks regression models.
After 1:1 propensity score matching, a total of 23 226 patients with newly diagnosed RA were equally subgrouped into acupuncture cohort or no-acupuncture cohort according to their use of acupuncture. The basic characteristics of these patients were similar. A lower cumulative incidence of ischaemic stroke was found in the acupuncture cohort (log-rank test, p<0.001; immortal time (period from initial diagnosis of RA to index date) 1065 days; mean number of acupuncture visits 9.83. In the end, 341 patients in the acupuncture cohort (5.95 per 1000 person-years) and 605 patients in the no-acupuncture cohort (12.4 per 1000 person-years) experienced ischaemic stroke (adjusted SHR 0.57, 95% CI 0.50 to 0.65). The advantage of lowering ischaemic stroke incidence through acupuncture therapy in RA patients was independent of sex, age, types of drugs used, and comorbidities.
The authors concluded that this study showed the beneficial effect of acupuncture in reducing the incidence of ischaemic stroke in patients with RA.
It seems obvious that the editors of ‘BMJ Open’, the peer reviewers of the study and the authors are unaware of the fact that the objective of such an investigeation is not to to demonstrate that acupuncture is beneficial but to test whether acupuncture is beneficial. Starting a study with the intention to to show that my pet therapy works is akin to saying: “I am intending to mislead you about the value of my intervention”.
One needs therefore not be surprised that the authors of the present study draw very definitive conclusions, such as “acupuncture therapy is beneficial for ischaemic stroke prevention”. But every 1st year medical or science student should know that correlation is not the same as causation. What the study does, in fact, show is an association between acupuncture and stroke. This association might be due to dozens of factors that the ‘propensity score matching’ could not control. To conclude that the results prove a cause effect relationship is naive bordering on scientific misconduct. I find it most disappointing that such a paper can pass all the hurdles to get published in what pretends to be a respectable journal.
Personally, I intend to use this study as a good example for drawing the wrong conclusions on seemingly rigorous research.
An article entitled “Homeopathy for worm infestations in children’s” caught my eye. Here is the un altered abstract:
Unusual sorts of worms can be there in a child’s stomach and may initiate several health complications such as pain, infection etc. To treat worm infections, one must identify about various categories of worms, and after understanding the kind of infection induced by the worm’s the treatment for the babies or children can planned. There are various Homeopathic medicines available which be used to treat worm infections without any side effects on the health of the children’s. In this paper we have discussed how the worm infection has been treated at our homeopathic research institute.
In the paper itself, the author, Dr. AK Dwivedi (Professor and H.O.D, Department of Physiology and Biochemistry, SKRP Guajarati Medical College, Indore, Madhya Pradesh, India), explains:
Homeopathic do not take care of just the disease, but is prescribed on the base of physical, emotional and genetic condition of a person. Homeopathic medicines act on both the mental and physical levels of individuals. Homeopathy is a natural and mild system of medicines that utilizes minute doses of well-researched remedies to improve the body’s natural curing procedure. Homeopathic medicines originate from substances that come’s from plants, minerals & animals.
The author’s conclusion is equally impressive:
On the basis of our research and after analysing the patients after medication we have found that homeopathy can completely cure the worm infection, If the dosages are properly prescribed and taken on that basis of investigation reports from time to time under guidance of experienced Homeopathic Physician worm infection can be treated with homeopathy effectively.
Yes, this level of incompetence could be quite funny! But sadly, it is also quite concerning. Most worm infestations are easily treated with effective conventional therapies. Untreated, they can have serious consequences. To advocate homeopathy – which is of course ineffective – is irresponsible, unethical and arguably criminal, in my view.