Earlier this year, I started the ‘WORST PAPER OF 2022 COMPETITION’. You will ask: what is there to win in this competition? I agree: a competition without a prize is no fun. Therefore, I suggest offering the winner (that is the author of the winning paper) one of my books that best fits his/her subject. I am sure this will overjoy him or her. And how do we identify the winner? I will I continue blogging about nominated papers (I hope to identify about 10 in total), and towards the end of the year, I let my readers decide democratically.
In this spirit of democratic voting, let me suggest to you ENTRY No 7:
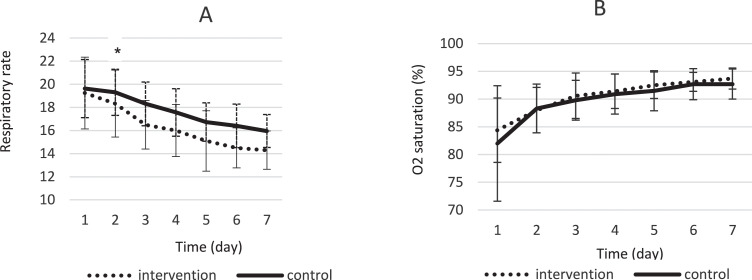
This trial evaluated the efficacy and safety of a Persian herbal medicine formula on patients with COVID-19. It was conducted in Afzalipour hospital, Kerman, Iran. Hospitalized COVID-19 patients were randomly divided into intervention (Persian herbal medicine formula + routine treatment) or control (only standard treatment) groups. The intervention group received capsule number 1 and 2 every 8 hours for 7 days. Capsule number 1 contained an extract of the Glycyrrhiza glabra, Punica granatum, and Rheum palmatum, and capsule number 2 was filled with Nigella sativa powder. Participants were followed up for 7 days. The primary outcome was the number of hospitalization days, while cough, fever, respiratory rate, days on oxygen (O2) therapy, and mortality rate were considered as secondary outcomes.
Eighty-two patients were enrolled in the study, while 79 cases completed the trial and their data were analyzed. The Persian medicine formula decreased the mean hospitalization days, so that the mean difference in length of hospitalization was 2.95 ± 0.43 days. A significant clinical improvement was observed regarding dyspnea, need for O2) therapy, and respiratory rate in the intervention group. Fever severity, cough severity, and death percentage were not statistically significantly different between groups. No adverse effects were reported.
The authors concluded that the present study supported the use of intended Persian medicine formulas as a supportive treatment for the hospitalized COVID-19 patients in order to improve their sign and symptoms, accelerate treatment, and shorten their hospitalization day during the disease period. It is recommended to conduct more clinical studies with bigger sample size for better evaluation.
This study begs a few questions:
- Why did the researchers not use a placebo in the control group?
- Why did they include only 82 patients in their study?
- Why did they not include outcomes that were independent of subjective influences?
- Why was mortality mentioned but no data provided?
- How is it possible that, under the conditions of the study, no adverse effects were noted?
- Why did you not stress the strong possibility that the effects are entirely due to placebo responses and wishful thinking?
- Why did you publish your study in a 3rd class journal?
I also have a few questions specifically for the editor of the Elsevier journal Integr Med Res, who decided to publish this study:
- Why did nobody bother to correct the poor English of the authors?
- Do you consider it ethical to publish such a poor-quality study?
- Are you not afraid that the conclusions might send many desperate patients up the garden path?
We are still in a pandemic where hundreds of people die every day. This means we are all extremely keen to find effective treatments. But it also means that an important ethical duty rests on all concerned to do rigorous research and abstain from unwarranted conclusions that might mislead us all.
The way I see it, there are two this paper can cause harm:
- Serious researchers will be put off from further investigating the herbal treatment. If the therapy is indeed effective, this would mean that progress is being hindered.
- More likely the treatment is useless. In this case, this study might cause considerable harm by prompting people to rely on an ineffective therapy.

Leave a Reply