Zinc has been in the limelight recently. The reason is that it has been recommended as a preventative and/or treatment of COVID infections. The basis for such recommendations has been some trial evidence suggesting it is effective for viral respiratory tract infections (RTIs). But the evidence has been full of contradictions which means, we need a systematic review that critically evaluated the totality of the available data.
This systematic review was aimed at evaluating the benefits and risks of zinc formulations compared with controls for the prevention or treatment of acute RTIs in adults.
Seventeen English and Chinese databases were searched in April/May 2020 for randomized clinical trials (RCTs), and from April/May 2020 to August 2020 for SARS-CoV-2 RCTs. Cochrane rapid review methods were applied. Quality appraisals used the Risk of Bias 2.0 and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach.
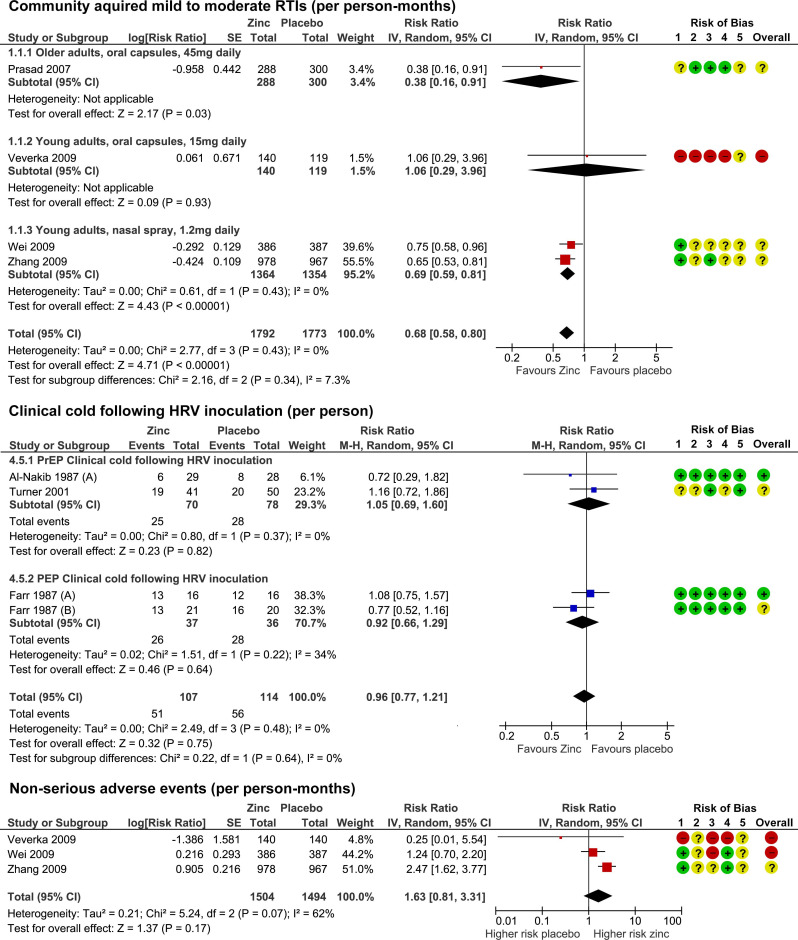
Twenty-eight RCTs with 5446 participants were identified. None were specific to SARS-CoV-2. Compared with placebo, oral or intranasal zinc prevented 5 RTIs per 100 person-months (95% CI 1 to 8, numbers needed to treat (NNT)=20, moderate-certainty/quality). Sublingual zinc did not prevent clinical colds following human rhinovirus inoculations (relative risk, RR 0.96, 95% CI 0.77 to 1.21, moderate-certainty/quality). On average, symptoms resolved 2 days earlier with sublingual or intranasal zinc compared with placebo (95% CI 0.61 to 3.50, very low-certainty/quality) and 19 more adults per 100 were likely to remain symptomatic on day 7 without zinc (95% CI 2 to 38, NNT=5, low-certainty/quality). There were clinically significant reductions in day 3 symptom severity scores (mean difference, MD -1.20 points, 95% CI -0.66 to -1.74, low-certainty/quality), but not average daily symptom severity scores (standardised MD -0.15, 95% CI -0.43 to 0.13, low-certainty/quality). Non-serious adverse events (AEs) (eg, nausea, mouth/nasal irritation) were higher (RR 1.41, 95% CI 1.17 to 1.69, NNHarm=7, moderate-certainty/quality). Compared with active controls, there were no differences in illness duration or AEs (low-certainty/quality). No serious AEs were reported in the 25 RCTs that monitored them (low-certainty/quality).
The authors concluded that in adult populations unlikely to be zinc deficient, there was some evidence suggesting zinc might prevent RTIs symptoms and shorten duration. Non-serious AEs may limit tolerability for some. The comparative efficacy/effectiveness of different zinc formulations and doses were unclear. The GRADE-certainty/quality of the evidence was limited by a high risk of bias, small sample sizes and/or heterogeneity. Further research, including SARS-CoV-2 clinical trials is warranted.
The authors provide a short comment on the assumed mode of action of zinc. The rationale for topical intranasal and sublingual zinc is based on the in vitro effects of zinc ions that can inhibit viral replication, stabilize cell membranes and reduce mucosal inflammation. Other conceivable mechanisms include the activation of T lymphocytes, monocytes, and granulocytes.
The authors also remind us to be cautious: clinicians and consumers need to be aware that considerable uncertainty remains regarding the clinical efficacy of different zinc formulations, doses, and administration routes, and the extent to which efficacy might be influenced by the ever changing epidemiology of the viruses that cause RTIs. The largest body of evidence comes from sublingual lozenges and zinc gluconate and acetate salts, suggesting these are suitable choices. Yet, this does not mean that other administration routes and zinc salts are less effective. The new evidence on the prophylactic effects of low-dose nasal sprays adds weight to the otherwise inconclusive findings from the handful of RCTs evaluating zinc nasal sprays or gels for acute treatment. A minimum therapeutic dose for zinc is also yet to be determined. An earlier review suggested the minimum dose for sublingual lozenges is 75 mg. However, the present analysis does not support this conclusion. Furthermore, a daily oral dose of 15 mg has been shown to upregulate lymphocytes within days, so it is plausible that much lower doses might also be effective.

As usual, conventional ‘medics’ and other assorted commentators have taken one nutrient out of context and discredited its importance. Even lay-people who conduct due diligence will appreciate that like many other minerals including selenium, magnesium, calcium, potassium etc etc it is wrong to even suggest that a single mineral acting in isolation can treat or cure any serious medical condition. This ‘expose’ of zinc is not worthy of being discussed on this forum or anywhere else for that matter. Vitamin D(3) has already been totally discredited by the UK Government and biased ‘experts.’
Vitamins, minerals, fats and proteins work together synergistically in the human body – and in the right balance they contribute to many important bodily functions that are vital to maintaining optimum health and ultimately our survival.
Who has “discredited its importance”?
@Michael Grant
I’m afraid that you miss the point of this research.
Um, a single mineral acting in isolation can treat and even cure a serious medical condition – the deficiency disease for that particular mineral, to be precise.
But this is not the point of the studies in this review. The point is that in vitro, zinc has measurable antiviral properties. There is also compelling evidence that zinc deficiency is associated with a worse outcome of Covid-19, and perhaps other viral infections as well, so there is definitely something about zinc and viral infections.
It is therefore a logical step to see if administering supplemental zinc has any influence on viral infections, and that is what was studied here.
Sure, all those essential micronutrients are indeed necessary to keep us alive (which is why they’re called essential in the first place). But for most of these chemicals, the actual blood and tissue levels are not critical at all as long as they are high enough. And they’re high enough in most people in our western world, so they do not need any supplements, even when they’re suffering from a viral infection – which is also supported by the fact that so far, there are no (combinations of) supplements that offer clear therapeutic benefits for non-deficient patients.
Taking supplements does not help cure any disease.
Richard says and claims:
“Sure, all those essential micronutrients are indeed necessary to keep us alive (which is why they’re called
essential in the first place)”.
“And they’re high enough in most people in our western world, so they do not need any supplements”.
“Taking supplements does not -help- cure any disease”.
But what is the reality.. Conservative estimates suggest that 25% of the world’s population is at risk of zinc
deficiency.
https://en.wikipedia.org/wiki/Zinc_deficiency
Zinc deficiency can be caused by a diet high in phytate-containing whole grains, foods grown in zinc deficient
soil, or processed foods containing little or no zinc.
28% of adults in England are obese and a further 36% are overweight
https://commonslibrary.parliament.uk/research-briefings/sn03336/
Richard and I come from NL, the situation in NL will not be much better..
The Impact of micronutrient deficiencies on obesity
https://academic.oup.com/nutritionreviews/article/67/10/559/1817341
The Malnutrition of Obesity: Micronutrient Deficiencies That Promote Diabetes
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3313629
“Essential micronutrients) are high enough in most people in our western world”
Can you believe it ??
Does this include the western world? I don’t think so.
Overweight people generally consume too much food – and common sense would say that by eating too much, they should at the very least get sufficient minerals and other essential nutrients. (Unless of course their diet is so unbalanced that it not only is rich in energy, but also lacks vitamins etcetera.)
Yes, no doubt some of those nutrients preferentially get stored in fat, and that could lower their blood concentration, but I never before encountered any claim that obese people are almost by definition suffering from deficiencies and should therefore take supplements – unless their diet really is so unhealthy as to be sorely lacking in that respect.
And even if obesity levels increase to ~30% of the population and ALL obese people have some micronutrient deficiency, then that still means that most people in the western world do not need supplements.
What I find is that it is mostly medically and scientifically incompetent quacks such as orthomolecular practitioners and naturopaths who claim that large swaths of the population need supplements.
Richard does not take the problem of micronutrient deficiencies seriously.
It is his interpretation of it.
The problem of micronutrient deficiencies also exists in children. This study reports on that.
Suboptimal Micronutrient Intake among Children in Europe:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4446765/
Isn’t it rather that certain non medics take a vitamin/mineral/whatever out of context and exaggerate its ability to treat certain health issues despite the lack of evidence. Just because a deficiency of some element of nutrition may cause problems there is no reason to believe an excess of it will cure or prevent any other condition. The result is usually just expensive piss and bigger profits for the supplement pushers.
But if all you are saying is “eat food or die” then I’ll happily agree.
Michael
These “biased ‘experts'” of whom you speak.
Who would you consider to be an “unbiased expert”?
Zinc is an amazing mineral. along with Mg and Mb, it is the most important mineral for our bodies, immune system and health, generally. copper blocks zinc. and we ALL have (hidden)copper overload. Cu overload is responsible for all new age dis-ease including cancer. so it makes sense that Zinc is the be all and end all. but i wouldn’t be buying the plastic wrapped stuff. it’s supporting over-breeding (i’ll leave u to work that one out!) and this planet’s destruction. Zinc/protein is found in abundance in greeny leafy veg such as all the asian veggies like bok choy and mizuna, along with broccoli, parsley and, of course, our most beloved complete food, the wheat grass juice. taken daily your hair won’t go grey (i’m 66 and mine’s still brown!) and you’ll still be surfing some of the world’s wildest waves (as does Kelly Slater – thirteen times world champ still winning Backdoor at 50!). like i learned a long time ago the strongest animal in the world doesn’t eat meat!
may I ask you: what is your professional background?
[it evidently is not in medicine or science]